Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Hiramoto, Masayuki; Kobayashi, Yasushi; Nakama, Shigeo; Mizutani, Kazuhiko*; Morita, Atsushi*
JAEA-Research 2008-001, 84 Pages, 2008/03
In this study, the following studies on the design method of multi-tunnels were performed. Study on the outbreak behavior of EDZ (Chapter 2) Study on the stability evaluation of the pillars (Chapter 3) In Chapter 2, analysis was carried out for "multi-tunnels model" that modeled the gallery group of a disposal panel scale. As a result, it is important to use multi-tunnels models for prediction evaluation of EDZ, because the outbreak behavior of EDZ was different from the model of the H12 report. In Chapter 3, a stability evaluation method of pillars was considered from two viewpoints of "pillar strength" and "pillar load" based on the similar structures. As a result, if numerical analysis by the multi-tunnels model was carried out, pillar strength and pillar load was estimated adequately. Therefore, a stability evaluation method of the pillars in a disposal institution was suggested.
 solutions
solutionsToyoshima, Atsushi; Haba, Hiromitsu*; Tsukada, Kazuaki; Asai, Masato; Akiyama, Kazuhiko*; Goto, Shinichi*; Ishii, Yasuo; Nishinaka, Ichiro; Sato, Tetsuya; Nagame, Yuichiro; et al.
Radiochimica Acta, 96(3), p.125 - 134, 2008/03
Times Cited Count:29 Percentile:84.99(Chemistry, Inorganic & Nuclear)Formation of an anionic fluoride-complex of element 104, rutherfordium (Rf) produced in the  Cm(
Cm( O,5n)
O,5n) Rf reaction was studied by an anion-exchange method based on an atom-at-a-time scale. It was found that the hexafluoro complex of Rf, [RfF
Rf reaction was studied by an anion-exchange method based on an atom-at-a-time scale. It was found that the hexafluoro complex of Rf, [RfF ]
] , was formed in the studied fluoride ion concentrations of 0.0005 - 0.013 M. Formation of [RfF
, was formed in the studied fluoride ion concentrations of 0.0005 - 0.013 M. Formation of [RfF ]
] was significantly different from that of the homologues Zr and Hf, [ZrF
was significantly different from that of the homologues Zr and Hf, [ZrF ]
] and [HfF
and [HfF ]
] ; the evaluated formation constant of [RfF
; the evaluated formation constant of [RfF ]
] is at least one-order of magnitude smaller than those of [ZrF
is at least one-order of magnitude smaller than those of [ZrF ]
] and [HfF
and [HfF ]
] .
.
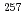 No and its
No and its  -decay daughter
-decay daughter 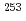 Fm
FmAsai, Masato; Tsukada, Kazuaki; Sakama, Minoru*; Ichikawa, Shinichi; Ishii, Tetsuro; Nagame, Yuichiro; Nishinaka, Ichiro; Akiyama, Kazuhiko; Osa, Akihiko; Oura, Yasuji*; et al.
Physical Review Letters, 95(10), p.102502_1 - 102502_4, 2005/09
Times Cited Count:63 Percentile:87.59(Physics, Multidisciplinary)Excited states in 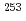 Fm fed by the
Fm fed by the  decay of
decay of 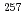 No have been established through
No have been established through  -
- and
and  -electron coincidence spectroscopy using a gas-jet transport system and an on-line isotope separator. The spin-parity of the 124.1 keV level in
-electron coincidence spectroscopy using a gas-jet transport system and an on-line isotope separator. The spin-parity of the 124.1 keV level in 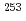 Fm has been identified on the basis of the measured internal conversion coefficients. The
Fm has been identified on the basis of the measured internal conversion coefficients. The ![$$nu 3/2^{+}[622]$$](/search/images/EROG23JAGMXTEXT1FN4VWNRSGJOSI.gif) configuration has been assigned to the ground state of
configuration has been assigned to the ground state of 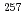 No as well as to the 124.1 keV level. It was found that the ground state configuration of
No as well as to the 124.1 keV level. It was found that the ground state configuration of 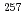 No is different from that of lighter
No is different from that of lighter 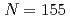 isotones.
isotones.
Nagame, Yuichiro; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Akiyama, Kazuhiko; Ishii, Yasuo; Sato, Tetsuya; Hirata, Masaru; Nishinaka, Ichiro; Ichikawa, Shinichi; et al.
Radiochimica Acta, 93(9-10), p.519 - 526, 2005/00
Times Cited Count:31 Percentile:87.03(Chemistry, Inorganic & Nuclear)no abstracts in English
Haba, Hiromitsu*; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Akiyama, Kazuhiko; Nishinaka, Ichiro; Hirata, Masaru; Yaita, Tsuyoshi; Ichikawa, Shinichi; Nagame, Yuichiro; et al.
Journal of the American Chemical Society, 126(16), p.5219 - 5224, 2004/04
Times Cited Count:44 Percentile:72.46(Chemistry, Multidisciplinary)Fluoride complexation of element 104, rutherfordium (Rf), produced in the  Cm(
Cm( O,5n)
O,5n) Rf reaction has been studied by anion-exchange chromatography on an atom-at-a-time scale. The anion-exchangechromatographic behavior of Rf was investigated in 1.9-13.9 M hydrofluoric acid together with those of the group-4 elements Zr and Hf produced in the
Rf reaction has been studied by anion-exchange chromatography on an atom-at-a-time scale. The anion-exchangechromatographic behavior of Rf was investigated in 1.9-13.9 M hydrofluoric acid together with those of the group-4 elements Zr and Hf produced in the  O-induced reactions on Ge and Gd targets, respectively. It was found that the adsorption behavior of Rf on anion-exchange resin is quite different from those of Zr and Hf, suggesting the influence of relativistic effect on the fluoride complexation of Rf.
O-induced reactions on Ge and Gd targets, respectively. It was found that the adsorption behavior of Rf on anion-exchange resin is quite different from those of Zr and Hf, suggesting the influence of relativistic effect on the fluoride complexation of Rf.
Nagame, Yuichiro; Haba, Hiromitsu*; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Goto, Shinichi*; Akiyama, Kazuhiko; Kaneko, Tetsuya; Sakama, Minoru*; Hirata, Masaru; et al.
Nuclear Physics A, 734, p.124 - 135, 2004/04
Times Cited Count:11 Percentile:56.87(Physics, Nuclear)no abstracts in English
 Bk
BkAsai, Masato; Tsukada, Kazuaki; Ichikawa, Shinichi; Sakama, Minoru*; Haba, Hiromitsu*; Nagame, Yuichiro; Nishinaka, Ichiro; Akiyama, Kazuhiko; Toyoshima, Atsushi; Kaneko, Tetsuya*; et al.
European Physical Journal A, 16(1), p.17 - 19, 2003/01
Times Cited Count:8 Percentile:50.24(Physics, Nuclear)The new isotope  Bk has been identified for the first time using a gas-jet coupled on-line isotope separator at the JAERI tandem accelerator facility. Characteristic X rays and three
Bk has been identified for the first time using a gas-jet coupled on-line isotope separator at the JAERI tandem accelerator facility. Characteristic X rays and three  transitions associated with the EC decay of
transitions associated with the EC decay of  Bk have been observed, and the half-life was determined to be 4.6 min. The half-life value and the observed
Bk have been observed, and the half-life was determined to be 4.6 min. The half-life value and the observed  -ray energies and intensities can be consistently explained as a consequence of the allowed EC transition of
-ray energies and intensities can be consistently explained as a consequence of the allowed EC transition of ![$$pi 7/2^{+}[633]$$](/search/images/EROHA0JAG2XTEXT1FN4VWNRTGNOSI.gif)

![$$nu 7/2^{+}[624]$$](/search/images/EROG23JAG2XTEXT1FN4VWNRSGROSI.gif) .
.
Asai, Masato; Sakama, Minoru*; Tsukada, Kazuaki; Ichikawa, Shinichi; Haba, Hiromitsu; Nishinaka, Ichiro; Nagame, Yuichiro; Goto, Shinichi*; Akiyama, Kazuhiko; Toyoshima, Atsushi; et al.
Journal of Nuclear and Radiochemical Sciences, 3(1), p.187 - 190, 2002/06
EC and  decays of neutron-deficient americium, curium, and berkelium nuclei have been studied using the JAERI on-line isotope separator. Decay schemes of
decays of neutron-deficient americium, curium, and berkelium nuclei have been studied using the JAERI on-line isotope separator. Decay schemes of 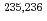 Am have been established for the first time, and proton-neutron configurations in
Am have been established for the first time, and proton-neutron configurations in 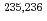 Am and
Am and 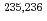 Pu have been determined.
Pu have been determined.  transitions observed in
transitions observed in 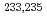 Am were found to be the favored transitions between the
Am were found to be the favored transitions between the ![$$pi 5/2^{-}[523]$$](/search/images/EROHA0JAGUXTEXT1FV4VWNJSGNOSI.gif) Nilsson states.
Nilsson states. 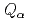 values of
values of  Cm have been determined. Nuclear structure, deformation, and atomic masses for these nuclei were discussed.
Cm have been determined. Nuclear structure, deformation, and atomic masses for these nuclei were discussed.
 solutions
solutionsHaba, Hiromitsu; Tsukada, Kazuaki; Asai, Masato; Goto, Shinichi*; Toyoshima, Atsushi; Nishinaka, Ichiro; Akiyama, Kazuhiko; Hirata, Masaru; Ichikawa, Shinichi; Nagame, Yuichiro; et al.
Journal of Nuclear and Radiochemical Sciences, 3(1), p.143 - 146, 2002/06
We have investigated the sorption behavior of element 104 rutherfordium (Rf) on an anion exchange resin from HCl and HNO solutions. In the HCl experiments, the distribution coefficients of Rf increase with an increase of HCl concentration from 7.0 M to 11.5 M, indicating that anionic species such as [Rf(OH)Cl
solutions. In the HCl experiments, the distribution coefficients of Rf increase with an increase of HCl concentration from 7.0 M to 11.5 M, indicating that anionic species such as [Rf(OH)Cl ]
] or [RfCl
or [RfCl ]
] are formed. This sorption behavior of Rf is typical of the group-4 elements Zr and Hf, and is quite different from that of the pseudo-homologue Th. It is also noted that the distribution coefficients decrease in the order Rf, Zr, Hf at 9.0 M HCl, which is consistent with the expected order of ionic radii. On the other hand, Rf appears to behave like Zr and Hf in 8 M HNO
are formed. This sorption behavior of Rf is typical of the group-4 elements Zr and Hf, and is quite different from that of the pseudo-homologue Th. It is also noted that the distribution coefficients decrease in the order Rf, Zr, Hf at 9.0 M HCl, which is consistent with the expected order of ionic radii. On the other hand, Rf appears to behave like Zr and Hf in 8 M HNO not like Pu and Th.
not like Pu and Th.
Nagame, Yuichiro; Asai, Masato; Haba, Hiromitsu; Tsukada, Kazuaki; Goto, Shinichi; Sakama, Minoru; Nishinaka, Ichiro; Toyoshima, Atsushi; Akiyama, Kazuhiko; Ichikawa, Shinichi
Journal of Nuclear and Radiochemical Sciences, 3(1), p.129 - 132, 2002/06
no abstracts in English
 Rf and
Rf and  Db in bombardments of
Db in bombardments of  Cm with
Cm with  O and
O and  F ions
F ionsNagame, Yuichiro; Asai, Masato; Haba, Hiromitsu; Goto, Shinichi*; Tsukada, Kazuaki; Nishinaka, Ichiro; Nishio, Katsuhisa; Ichikawa, Shinichi; Toyoshima, Atsushi*; Akiyama, Kazuhiko*; et al.
Journal of Nuclear and Radiochemical Sciences, 3(1), p.85 - 88, 2002/06
no abstracts in English
 -ray irradiation
-ray irradiationHosoi, Fumio; ; Akama, Kazuhiko*; *; *; *
J. Jpn. Oil Chem. Soc., 47(1), p.31 - 40, 1998/00
no abstracts in English
 -ray irradiation and its application
-ray irradiation and its applicationHosoi, Fumio; Akama, Kazuhiko*
Hyomen, 35(11), p.598 - 609, 1997/00
no abstracts in English
Hosoi, Fumio; Omichi, Hideki; Akama, Kazuhiko*; *; *; *
Nuclear Instruments and Methods in Physics Research B, 131(1-4), p.329 - 334, 1997/00
Times Cited Count:9 Percentile:59.74(Instruments & Instrumentation)no abstracts in English
Hosoi, Fumio; Omichi, Hideki; Akama, Kazuhiko*; Awai, K.*; Tokuyama, S.*; Sato, T.*
Nuclear Instruments and Methods in Physics Research B, 105(1-4), p.318 - 321, 1995/11
Times Cited Count:1 Percentile:23.9(Instruments & Instrumentation)no abstracts in English
 -radiation induced polymerization of unsaturated liposomes containing unsaturated lipid cholesterol, and saturated aliphatic acid
-radiation induced polymerization of unsaturated liposomes containing unsaturated lipid cholesterol, and saturated aliphatic acidHosoi, Fumio; Omichi, Hideki; Akama, Kazuhiko*; Tokuyama, S.*; Nakano, Y.*
JAERI-Conf 95-003, p.507 - 511, 1995/03
no abstracts in English
Tsukada, Kazuaki; Toyoshima, Atsushi; Haba, Hiromitsu*; Asai, Masato; Akiyama, Kazuhiko*; Ishii, Yasuo; Tome, Hayato*; Nishinaka, Ichiro; Sato, Tetsuya; Ichikawa, Takatoshi; et al.
no journal, ,
no abstracts in English
 OH mixed solution
OH mixed solutionTsukada, Kazuaki; Toyoshima, Atsushi; Haba, Hiromitsu*; Asai, Masato; Akiyama, Kazuhiko*; Ishii, Yasuo; Tome, Hayato; Nishinaka, Ichiro; Sato, Tetsuya; Ichikawa, Shinichi; et al.
no journal, ,
no abstracts in English
Nagame, Yuichiro; Toyoshima, Atsushi; Ishii, Yasuo; Tsukada, Kazuaki; Asai, Masato; Tome, Hayato; Kasamatsu, Yoshitaka; Nishinaka, Ichiro; Sato, Tetsuya; Haba, Hiromitsu*; et al.
no journal, ,
We present a study of fluoride complex formation of Rf at Japan Atomic Energy Agency (JAEA). Anion- and cation-exchange behavior of Rf was systematically investigated in HF/HNO mixed solution together with the lighter homologues Zr and Hf using the automated ion-exchange separation apparatus coupled with the detection system for alpha spectroscopy. We unequivocally determined the species of Rf on the binding sites of the resin as the hexafluoro complex [RfF
mixed solution together with the lighter homologues Zr and Hf using the automated ion-exchange separation apparatus coupled with the detection system for alpha spectroscopy. We unequivocally determined the species of Rf on the binding sites of the resin as the hexafluoro complex [RfF ]
] . It is found that there is about two-orders of magnitude difference in the fluoride ion concentration between Rf and the homologues for the formation of the hexafluoro complexes. The result clearly demonstrates that the formation of the fluoride complexes of Rf is much weaker than those of the homologues Zr and Hf.
. It is found that there is about two-orders of magnitude difference in the fluoride ion concentration between Rf and the homologues for the formation of the hexafluoro complexes. The result clearly demonstrates that the formation of the fluoride complexes of Rf is much weaker than those of the homologues Zr and Hf.
 media
mediaKasamatsu, Yoshitaka; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Haba, Hiromitsu*; Ishii, Yasuo; Tome, Hayato; Nishinaka, Ichiro; Akiyama, Kazuhiko*; Kikunaga, Hidetoshi*; et al.
no journal, ,
Dubnium-262 was produced in the  Cm(
Cm( F, 5
F, 5 ) reaction at the JAEA tandem accelerator. Adsorption of Db on the anion-exchange resin was investigated in 0.89 M HF/0.3 M HNO
) reaction at the JAEA tandem accelerator. Adsorption of Db on the anion-exchange resin was investigated in 0.89 M HF/0.3 M HNO solution. The anion-exchange behavior of Nb, Ta, and Pa as homologues of Db was also examined in details in HF/HNO
solution. The anion-exchange behavior of Nb, Ta, and Pa as homologues of Db was also examined in details in HF/HNO solutions. From the comparison of those results, we found that the adsorption of Db on the anion-exchange resin is considerably weaker than that of Ta and is relatively similar to those of Nb and Pa in the studied conditions.
solutions. From the comparison of those results, we found that the adsorption of Db on the anion-exchange resin is considerably weaker than that of Ta and is relatively similar to those of Nb and Pa in the studied conditions.