Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ohshima, Hiroyuki; Morishita, Masaki*; Aizawa, Kosuke; Ando, Masanori; Ashida, Takashi; Chikazawa, Yoshitaka; Doda, Norihiro; Enuma, Yasuhiro; Ezure, Toshiki; Fukano, Yoshitaka; et al.
Sodium-cooled Fast Reactors; JSME Series in Thermal and Nuclear Power Generation, Vol.3, 631 Pages, 2022/07
This book is a collection of the past experience of design, construction, and operation of two reactors, the latest knowledge and technology for SFR designs, and the future prospects of SFR development in Japan. It is intended to provide the perspective and the relevant knowledge to enable readers to become more familiar with SFR technology.
 and Eu
and Eu ions onto sedimentary rock in the presence of gamma-irradiated humic acid
ions onto sedimentary rock in the presence of gamma-irradiated humic acidZhao, Q.*; Saito, Takeshi*; Miyakawa, Kazuya; Sasamoto, Hiroshi; Kobayashi, Taishi*; Sasaki, Takayuki*
Journal of Hazardous Materials, 428, p.128211_1 - 128211_10, 2022/04
Times Cited Count:5 Percentile:62.11(Engineering, Environmental)The influence of humic acid and its radiological degradation on the sorption of Cs and Eu
and Eu by sedimentary rock was investigated to understand the sorption process of metal ions and humic substances. Aldrich humic acid (HA) solution was irradiated with different doses of gamma irradiation using a Co-60 gamma-ray source prior to the contact between the metal ions and the solid sorbent. The HA molecule decomposed to smaller molecules with a lower complexation affinity. Batch sorption experiments were performed to evaluate the effect of gamma-irradiated HA on the sorption of Cs
by sedimentary rock was investigated to understand the sorption process of metal ions and humic substances. Aldrich humic acid (HA) solution was irradiated with different doses of gamma irradiation using a Co-60 gamma-ray source prior to the contact between the metal ions and the solid sorbent. The HA molecule decomposed to smaller molecules with a lower complexation affinity. Batch sorption experiments were performed to evaluate the effect of gamma-irradiated HA on the sorption of Cs and Eu
and Eu ions. The addition of non-irradiated HA weakened the sorption of Eu because of the lower sorption of the neutral or negatively charged Eu-HA complexes compared with free Eu ions. The sorption of monovalent Cs ions was barely affected by the presence of HA and its gamma irradiation. The concentration ratio of HA complexed species and non-complexed species in the solid and liquid phases was evaluated by sequential filtration and chemical equilibrium calculations. The ratios supported the minimal contribution of HA to Cs sorption. However, the concentration ratio for Eu
ions. The addition of non-irradiated HA weakened the sorption of Eu because of the lower sorption of the neutral or negatively charged Eu-HA complexes compared with free Eu ions. The sorption of monovalent Cs ions was barely affected by the presence of HA and its gamma irradiation. The concentration ratio of HA complexed species and non-complexed species in the solid and liquid phases was evaluated by sequential filtration and chemical equilibrium calculations. The ratios supported the minimal contribution of HA to Cs sorption. However, the concentration ratio for Eu in the liquid phase was high, indicating that the complexing ability of HA to Eu
in the liquid phase was high, indicating that the complexing ability of HA to Eu was higher than that of HA to Cs
was higher than that of HA to Cs ions. Therefore, the sorption of free Eu
ions. Therefore, the sorption of free Eu would predominate with the gamma irradiation dose applied to the HA solution under a radiation field near the HLW package.
would predominate with the gamma irradiation dose applied to the HA solution under a radiation field near the HLW package.
Dohi, Terumi; Omura, Yoshihito*; Yoshimura, Kazuya; Sasaki, Takayuki*; Fujiwara, Kenso; Kanaizuka, Seiichi*; Nakama, Shigeo; Iijima, Kazuki
PLOS ONE (Internet), 16(5), p.e0251828_1 - e0251828_16, 2021/05
Times Cited Count:6 Percentile:41.49(Multidisciplinary Sciences)Nagao, Fumiya; Niizato, Tadafumi; Sasaki, Yoshito; Ito, Satomi; Watanabe, Takayoshi; Dohi, Terumi; Nakanishi, Takahiro; Sakuma, Kazuyuki; Hagiwara, Hiroki; Funaki, Hironori; et al.
JAEA-Research 2020-007, 249 Pages, 2020/10
The accident of the Fukushima Daiichi Nuclear Power Station, Tokyo Electric Power Company Holdings, Inc. occurred due to the Great East Japan Earthquake, Sanriku offshore earthquake, of 9.0 magnitude and the accompanying tsunami. As a result, large amount of radioactive materials was released into the environment. Under these circumstances, Japan Atomic Energy Agency (JAEA) has been conducting "Long-term Assessment of Transport of Radioactive Contaminants in the Environment of Fukushima" concerning radioactive materials released in environment, especially migration behavior of radioactive cesium since November 2012. This report is a summary of the research results that have been obtained in environmental dynamics research conducted by JAEA in Fukushima Prefecture.
Nagao, Fumiya; Niizato, Tadafumi; Sasaki, Yoshito; Ito, Satomi; Watanabe, Takayoshi; Dohi, Terumi; Nakanishi, Takahiro; Sakuma, Kazuyuki; Hagiwara, Hiroki; Funaki, Hironori; et al.
JAEA-Research 2019-002, 235 Pages, 2019/08
The accident of the Fukushima Daiichi Nuclear Power Station (hereinafter referred to 1F), Tokyo Electric Power Company Holdings, Inc. occurred due to the Great East Japan Earthquake, Sanriku offshore earthquake, of 9.0 magnitude and the accompanying tsunami. As a result, large amount of radioactive materials was released into the environment. Under these circumstances, JAEA has been conducting Long-term Environmental Dynamics Research concerning radioactive materials released in environment, especially migration behavior of radioactive cesium since November 2012. This report is a summary of the research results that have been obtained in environmental dynamics research conducted by JAEA in Fukushima Prefecture.
Funaki, Hironori; Takahara, Shogo; Sasaki, Miyuki; Yoshimura, Kazuya; Nakama, Shigeo; Sanada, Yukihisa
JAEA-Research 2018-016, 48 Pages, 2019/03
Cabinet Office Nuclear Emergency Response Headquarters starts to consider radiation protection in the "specific reconstruction reproduction base area" of which evacuation order will be lifted by 2023. It is essential to grab the present situations of radiation contamination and evaluate exposure dose in the area to realize the plan. Many surveys have evaluated the distributions of air dose rate and exposure dose has been estimated based on the results since the Fukushima Daiichi Nuclear Power Plant accident. Nevertheless, more detailed information on exposure is needed for the areas because its radiation level is relatively high. That is also to help make prudent evaluation plan. This study aimed to evaluate the detailed contamination situation there and estimate exposure dose with considering areal circumstances. Investigations were carried out for (1) airborne survey of air dose rate using an unmanned helicopter (2) evaluation of airborne radiocesium and (3) estimation of external/internal effective doses for typical activity patterns assumed. Additionally, we applied new methods for the airborne survey to evaluate exposure dose. Our study showed a detailed three-dimensional map of air dose rate and clarified the distribution pattern in the areas. Results of effective dose estimation suggested that the internal effective dose due to inhalation accounts for less than 1% of the external effective dose.
Yu, Q.*; Onuki, Toshihiko*; Kozai, Naofumi; Sakamoto, Fuminori; Tanaka, Kazuya; Sasaki, Keiko*
Chemical Geology, 470, p.141 - 151, 2017/10
Times Cited Count:15 Percentile:55.72(Geochemistry & Geophysics)In this work, the Cs retention onto two types of Mn oxide was investigated. We found that Todorokite has sorption sites with a higher selectivity for Cs than birnessite. When the initial Cs concentration was 10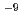 mol/L for the sorption experiments, approximately 34% of the sorbed Cs was residual in the todorokite after the extraction using 1 M NaCl and NH
mol/L for the sorption experiments, approximately 34% of the sorbed Cs was residual in the todorokite after the extraction using 1 M NaCl and NH Cl; this value was much higher than the results for the Cs-sorbed birnessite. These results strongly suggest that todorokite contributes to the fixation of radioactive Cs in soils.
Cl; this value was much higher than the results for the Cs-sorbed birnessite. These results strongly suggest that todorokite contributes to the fixation of radioactive Cs in soils.
Sasaki, Takayuki*; Kokami, Takayuki*; Kobayashi, Taishi*; Kirishima, Akira*; Murakami, Hiroaki; Amano, Yuki; Mizuno, Takashi; Iwatsuki, Teruki; Sasamoto, Hiroshi; Miyakawa, Kazuya
Journal of Nuclear Science and Technology, 54(3), p.373 - 381, 2017/03
Trace amounts of natural thorium and uranium in deep groundwater were investigated at two underground research laboratories situated at Horonobe and Mizunami, Japan. The groundwater was sampled from underground boreholes, and the colloid contribution was checked by in situ two size-fractionated ultrafiltration systems. A decrease in the concentration after in situ filtration suggested the presence of natural colloids and suspended matter that were carriers of a portion of the elements. The result of the Th and U concentrations in groundwater after 10 kDa filtration was analyzed thermodynamically using existing hydrogeological and geochemical data such as the mineral components in the groundwater at a given pH, ionic strength, concentration of co-existing ions, redox potential, and solid phase assumed. A crystalline solid phase made the solubility very low compared with that of the amorphous phase, and the solubility agreed well with the concentrations measured.
Kirishima, Akira*; Kuno, Atsushi*; Amamiya, Hiroki; Kubota, Takumi*; Kimuro, Shingo*; Amano, Yuki; Miyakawa, Kazuya; Iwatsuki, Teruki; Mizuno, Takashi; Sasaki, Takayuki*; et al.
Chemosphere, 168, p.798 - 806, 2017/02
Times Cited Count:3 Percentile:10.21(Environmental Sciences)For better understanding of the migration behavior of minor actinides (MA) in deep groundwater, the interaction of doped rare earth elements (REEs) and components in Horonobe deep groundwater was studied. Appx. 10 ppb of rare earth elements, i.e., Y, La, Ce, Pr, Nd, Sm, Eu, Gd, Tb, Dy, Er, Tm and Yb were doped to the sample groundwater collected from a packed sections in borehole drilled from 140 m depth experiment drift of Horonobe underground research laboratory (URL), Hokkaido, Japan. Then, that groundwater was sequentially filtrated by 0.2 micron pore filter, 10 kDa, 3 kDa and 1 kDa of nominal molecular weight limit (NMWL) ultrafilters by keeping inert condition. After that, the filtrate solutions were analyzed by ICP-MS to determine the concentrations of retained REEs at each filtration steps, while the used filters were analyzed by the neutron activation analysis (NAA) and TOF-SIMS element mapping to know the amount and chemical speciation of trapped fraction of the REEs on each filter. A remarkable relation between the retention ratios of REEs in the filtrate solutions and the ionic radius was observed, i.e., smaller rare earth element solves more in liquid phase under the Horonobe groundwater condition. NAA and TOF-SIMS analyses revealed that certain portions of REEs were trapped by 0.2 micron pore filters as rare earth phosphates which corresponded with the predicted predominant species by a chemical equilibrium calculation for the Horonobe groundwater condition, while small portions of colloidal REEs were trapped by 10 kDa and 3 kDa NMWL ultrafilters. The result suggested that phosphate anion plays an important role in the chemical behavior of REEs in saline (seawater based) groundwater, which could be referred for the prediction of migration behavior of trivalent actinide released from the repository of radioactive waste in far future.
 generation of two strains of
generation of two strains of 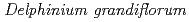 var.
var. 
Honda, Kazushige*; Taneichi, Shuhei*; Maeda, Tomoo*; Goto, Satoshi*; Shikanai, Yasuhiro*; Sasaki, Kazuya*; Nozawa, Shigeki; Hase, Yoshihiro
JAEA-Review 2015-022, JAEA Takasaki Annual Report 2014, P. 107, 2016/02
no abstracts in English
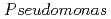 sp. strain NGY-1
sp. strain NGY-1Tanaka, Kazuya*; Yu, Q.; Sasaki, Keiko*; Onuki, Toshihiko
Geomicrobiology Journal, 30(10), p.874 - 885, 2013/08
Times Cited Count:15 Percentile:41.01(Environmental Sciences)no abstracts in English
Yu, Q.; Sasaki, Keiko*; Tanaka, Kazuya*; Onuki, Toshihiko; Hirajima, Tsuyoshi*
Geomicrobiology Journal, 30(9), p.829 - 839, 2013/07
Times Cited Count:35 Percentile:71.68(Environmental Sciences)no abstracts in English
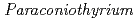 sp. WL-2 strain affecting sorption of Co
sp. WL-2 strain affecting sorption of Co
Yu, Q.*; Sasaki, Keiko*; Tanaka, Kazuya*; Onuki, Toshihiko; Hirajima, Tsuyoshi*
Chemical Geology, 310-311, p.106 - 113, 2012/06
Times Cited Count:63 Percentile:84.96(Geochemistry & Geophysics)These results strongly suggested that the interlayer Mn(III) can oxidize the adsorbed Co to Co
to Co , resulting in specific adsorption of Co
, resulting in specific adsorption of Co by biogenic birnessite.
by biogenic birnessite.
Hoshino, Tsuyoshi; Kato, Kenichi*; Natori, Yuri*; Oikawa, Fumiaki; Nakano, Natsuko*; Nakamura, Mutsumi*; Sasaki, Kazuya*; Suzuki, Akihiro*; Terai, Takayuki*; Tatenuma, Katsuyoshi*
Journal of Nuclear Materials, 417(1-3), p.684 - 687, 2011/10
Times Cited Count:52 Percentile:96.13(Materials Science, Multidisciplinary)Lithium titanate (Li TiO
TiO ) is one of the most promising candidates among tritium breeding materials because of its good tritium release. Addition of H
) is one of the most promising candidates among tritium breeding materials because of its good tritium release. Addition of H to inert sweep gas has been proposed for enhancing the tritium release from tritium breeding materials. However, the mass of Li
to inert sweep gas has been proposed for enhancing the tritium release from tritium breeding materials. However, the mass of Li TiO
TiO was decreased with time in the hydrogen atmosphere. It is assumed that the mass decrease indicates the loss of the oxygen contained in the sample caused by the change from Ti
was decreased with time in the hydrogen atmosphere. It is assumed that the mass decrease indicates the loss of the oxygen contained in the sample caused by the change from Ti  to Ti
to Ti  , and that the partial pressures of Li-containing species were increased in the hydrogen atmosphere. In order to decrease the mass-change at high temperature, advanced tritium breeding material with added Li should be developed to improve the physical and chemical stability in hydrogen atmosphere. In the case of the Li
, and that the partial pressures of Li-containing species were increased in the hydrogen atmosphere. In order to decrease the mass-change at high temperature, advanced tritium breeding material with added Li should be developed to improve the physical and chemical stability in hydrogen atmosphere. In the case of the Li TiO
TiO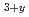 samples used by the present study, LiOHH
samples used by the present study, LiOHH O and H
O and H TiO
TiO were proportionally mixed with the molar ratio Li/Ti of either 2.0 and 2.2. These samples are designated as L20 (Li/Ti = 2.0) and L22 (Li/Ti = 2.2), respectively. The results of XRD measurement showed that the phases in advanced tritium breeding material were as follows. L22 existed as non-stoichiometric compound Li
were proportionally mixed with the molar ratio Li/Ti of either 2.0 and 2.2. These samples are designated as L20 (Li/Ti = 2.0) and L22 (Li/Ti = 2.2), respectively. The results of XRD measurement showed that the phases in advanced tritium breeding material were as follows. L22 existed as non-stoichiometric compound Li TiO
TiO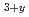 .
.
Hoshiya, Taiji; Nishimura, Akihiko; Sasaki, Kazuya*; Nishikawa, Masahiro*
Enerugi Kankyo Kyoiku Kenkyu, 4(2), p.49 - 56, 2010/07
The Kids' Science Museum of Photons (KSMP), which was established in a blanch of the Kansai Photon Science Institute (KPSI), can provide us with the mysteries of light through various exhibits, theater, and science experimental events, as quite an unique museum, to be focused on the photon science. The goal of this museum is also to develop "Heart of Science" in children who can join and experience. In this phase, the KSMP has been promoting various activities of public understanding of science and technology, such as the collaboration seminar, the science festival and the teacher education program, to be associated with regional administrative offices, educational institutions, and schools. It can be explained the outline of systematically trial activities on the energy and environmental education, the collaboration lecture, the experimental village and the science-walker, from the viewpoint of the degree of understanding, the search mind and effects.
Kofuji, Hirohide; Amamoto, Ippei; Sasaki, Kazuya*; Yasumoto, Masaru*; Takasaki, Yasushi*; Myochin, Munetaka; Terai, Takayuki*
Denki Kagaku Oyobi Kogyo Butsuri Kagaku, 77(8), p.597 - 600, 2009/08
Times Cited Count:0 Percentile:0.01(Electrochemistry)The process flow of the phosphate conversion technique has been developed for the reduction of high-level radioactive waste (HLW) generated from the metal-electrorefining process. In this study, the results of thermodynamic calculations for the phosphate conversion reactions were examined by the basic experiments. The chlorides of rare earth elements (REE) turned out to be converted into phosphates easily. Furthermore, as the additive for the phosphate conversion reaction, high temperature behavior of lithium phosphate was evaluated to elucidate the thermodynamic property.
 TiO
TiO additives for ITER-TBM
additives for ITER-TBMHoshino, Tsuyoshi; Kato, Kenichi*; Natori, Yuri*; Nakamura, Mutsumi*; Sasaki, Kazuya*; Hayashi, Kimio; Terai, Takayuki*; Tatenuma, Katsuyoshi*
Fusion Engineering and Design, 84(2-6), p.956 - 959, 2009/06
Times Cited Count:45 Percentile:93.9(Nuclear Science & Technology)Li TiO
TiO is one of the most promising candidates among the proposed solid breeder materials for fusion reactors. Addition of H
is one of the most promising candidates among the proposed solid breeder materials for fusion reactors. Addition of H to inert sweep gas has been proposed for enhancing the release of bred tritium from breeder material. However, the mass of Li
to inert sweep gas has been proposed for enhancing the release of bred tritium from breeder material. However, the mass of Li TiO
TiO was found to decrease with time in the hydrogen atmosphere. This mass change indicates that the oxygen content of the sample decreased, suggesting the change from Ti
was found to decrease with time in the hydrogen atmosphere. This mass change indicates that the oxygen content of the sample decreased, suggesting the change from Ti to Ti
to Ti . In order to control the mass change at the time of high temperature use, the development of lithium titanate which has Li
. In order to control the mass change at the time of high temperature use, the development of lithium titanate which has Li TiO
TiO additive is expected to be effective.
additive is expected to be effective.
Kofuji, Hirohide; Amamoto, Ippei; Yasumoto, Masaru*; Sasaki, Kazuya*; Myochin, Munetaka; Terai, Takayuki*
Journal of Nuclear Materials, 389(1), p.173 - 178, 2009/05
Times Cited Count:4 Percentile:30.49(Materials Science, Multidisciplinary)Adaptation of the phosphate conversion technique was undertaken and evaluated for application to the recycle process of the spent electrolyte generated from metal electrorefining process which is a kind of pyrochemical reprocessing technologies. The conceptual flow sheet has been discussed based on the thermodynamic properties of constituent. However few data have been published relating to the phosphate, therefore, the thermodynamic data were attempted to be obtained by the calorimetry and vapor pressure measurements. The measurements have been started with pure substance such as lithium phosphate. Through the measurements of HTMS and TG-DTA, it was clarified that decomposition or phase transition of Li PO
PO occurred at specific temperatures below the melting point of Li
occurred at specific temperatures below the melting point of Li PO
PO . To avoid the influence of fragment substances which have high vapour pressures, improved method of the HTMS measurement was examined.
. To avoid the influence of fragment substances which have high vapour pressures, improved method of the HTMS measurement was examined.
Hoshino, Tsuyoshi; Sasaki, Kazuya*; Tsuchiya, Kunihiko; Hayashi, Kimio; Suzuki, Akihiro*; Hashimoto, Takuya*; Terai, Takayuki*
Journal of Nuclear Materials, 386-388, p.1098 - 1101, 2009/04
no abstracts in English
Hoshiya, Taiji; Sasaki, Kazuya*; Nishikawa, Masahiro
Reza Kenkyu, 36(4), p.221 - 225, 2008/04
The Kansai Photon Science Institute (KPSI) of Japan Atomic Energy Agency (JAEA) has been promoting various activities of public understanding of science and technology, to be focused on the photon science, which is characterized by activities on science lectures and science events based on science and experiment classrooms, by utilizing the science museum of the JAEA (The Kids' Science Museum of Photons). In this phase, the KPSI extends systematically trial activities of the science museum to be as an experimental apparatus for studying on science and technology, including the program for promoting activities on public understanding of science and technology at the region. In this report, the photons rainbow-colored energy experiments lecture schools were held on the energy conversion. About ninety percent of 142 participants can enjoy each school and get the motivation to have attraction for these fields, and these schools can be useful for elementary school members.