Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ikeuchi, Hirotomo; Sasaki, Shinji; Onishi, Takashi; Nakayoshi, Akira; Arai, Yoichi; Sato, Takumi; Ohgi, Hiroshi; Sekio, Yoshihiro; Yamaguchi, Yukako; Morishita, Kazuki; et al.
JAEA-Data/Code 2023-005, 418 Pages, 2023/12
For safe and steady decommissioning of Tokyo Electric Power Company Holdings' Fukushima Daiichi Nuclear Power Station (1F), information concerning composition and physical/chemical properties of fuel debris generated in the reactors should be estimated and provided to other projects conducting the decommissioning work including the retrieval of fuel debris and the subsequent storage. For this purpose, in FY2021, samples of contaminants (the wiped smear samples and the deposits) obtained through the internal investigation of the 1F Unit 2 were analyzed to clarify the components and to characterize the micro-particles containing uranium originated from fuel (U-bearing particles) in detail. This report summarized the results of analyses performed in FY2021, including the microscopic analysis by SEM and TEM, radiation analysis, and elemental analysis by ICP-MS, as a database for evaluating the main features of each sample and the probable formation mechanism of the U-bearing particles.
Sudo, Ayako; M sz
sz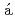 ros, B.*; Sato, Takumi; Nagae, Yuji
ros, B.*; Sato, Takumi; Nagae, Yuji
JAEA-Research 2023-007, 31 Pages, 2023/11
For the criticality assessment of fuel debris generated by the accident in Fukushima Daiichi Nuclear Power Station, understanding of the elemental localization in fuel debris is important. Especially, the distribution of Fe and Gd, which may behave as potential neutron absorber materials in the fuel debris, is of particular important from the viewpoint of nuclear criticality safety. To investigate the localization tendency of Gd and Fe in molten core materials during solidification progress, liquefaction/solidification tests on core materials containing UO , ZrO
, ZrO , FeO, Gd
, FeO, Gd O
O , and simulated fission products (MoO
, and simulated fission products (MoO , Nd
, Nd O
O , SrO, and RuO
, SrO, and RuO ) and concrete (SiO
) and concrete (SiO , Al
, Al O
O , and CaO) were performed using cold crucible induction heating technique. During the test, the molten core materials gradually subsided and solidified from the bottom to the top of the melt. Elemental analysis showed that Fe content in the inner region increased approximately up to 3.4 times that in the bottom region. The concentration of Fe into the inner region was observed in all the samples regardless of the initial FeO composition, cooling rates, and phase separation. This suggests that FeO may be concentrated into the low temperature region, where the melt solidified later. In contrast, Gd content in the bottom region increased approximately up to 2.6 times that in the inner region. The concentration of Gd into the bottom region was observed when the initial Gd
, and CaO) were performed using cold crucible induction heating technique. During the test, the molten core materials gradually subsided and solidified from the bottom to the top of the melt. Elemental analysis showed that Fe content in the inner region increased approximately up to 3.4 times that in the bottom region. The concentration of Fe into the inner region was observed in all the samples regardless of the initial FeO composition, cooling rates, and phase separation. This suggests that FeO may be concentrated into the low temperature region, where the melt solidified later. In contrast, Gd content in the bottom region increased approximately up to 2.6 times that in the inner region. The concentration of Gd into the bottom region was observed when the initial Gd O
O content was higher than 1 at.%. This suggests that Gd
content was higher than 1 at.%. This suggests that Gd O
O may be concentrated into the earlier solidified region. On the other hand, no significant localization was observed on the simulated fission products.
may be concentrated into the earlier solidified region. On the other hand, no significant localization was observed on the simulated fission products.
Watanabe, So; Takahatake, Yoko; Ogi, Hiromichi*; Osugi, Takeshi; Taniguchi, Takumi; Sato, Junya; Arai, Tsuyoshi*; Kajinami, Akihiko*
Journal of Nuclear Materials, 585, p.154610_1 - 154610_6, 2023/11
Times Cited Count:0 Percentile:0.01(Materials Science, Multidisciplinary)Sato, Takumi; Otobe, Haruyoshi; Morishita, Kazuki; Marufuji, Takato; Ishikawa, Takashi; Fujishima, Tadatsune; Nakano, Tomoyuki
JAEA-Technology 2023-016, 41 Pages, 2023/09
This report summarizes the results of the stabilization treatments of post-experiment nuclear materials in Plutonium Fuel Research Facility (PFRF) from August 2018 to March 2021. Based on the management standards for nuclear materials enacted after the contamination accident that occurred at PFRF on June 6, 2017, the post-experiment nuclear materials containing plutonium (Pu): samples mixed with organic substances that cause an increase in internal pressure due to radiolysis (including X-ray diffraction samples mixed with epoxy resin and plutonium powder which caused contamination accidents), carbides and nitrides samples which is reactive in air, and chloride samples which may cause corrosion of storage containers, were selected as targets of the stabilization. The samples containing organic materials, carbides and nitrides were heated in an air flow at 650  C and 950
C and 950  C for 2 hours respectively to remove organic materials and convert uranium (U) and Pu into oxides. U and Pu chlorides in LiCl-KCl eutectic melt were reduced and extracted into liquid Cd metal by a reaction with lithium (Li) -cadmium (Cd) alloy and converted to U-Pu-Cd alloy at 500
C for 2 hours respectively to remove organic materials and convert uranium (U) and Pu into oxides. U and Pu chlorides in LiCl-KCl eutectic melt were reduced and extracted into liquid Cd metal by a reaction with lithium (Li) -cadmium (Cd) alloy and converted to U-Pu-Cd alloy at 500  C or higher. All of the samples were stabilized and stored at PFRF. We hope that the contents of this report will be utilized to consider methods for stabilizing post experiment nuclear materials at other nuclear fuel material usage facilities.
C or higher. All of the samples were stabilized and stored at PFRF. We hope that the contents of this report will be utilized to consider methods for stabilizing post experiment nuclear materials at other nuclear fuel material usage facilities.
Yamashita, Susumu; Sato, Takumi; Nagae, Yuji; Kurata, Masaki; Yoshida, Hiroyuki
Journal of Nuclear Science and Technology, 60(9), p.1029 - 1045, 2023/09
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology)Sato, Yuji; Miyamoto, Yuta; Awatani, Yuto; Yamamoto, Kosuke; Hatakeyama, Takumi
JAEA-Review 2023-002, 59 Pages, 2023/08
"Fugen Decommissioning Engineering Center", in planning and carrying out our decommissioning technical development, organizes "Technical special committee on Fugen decommissioning" which consists of the members well-informed, aiming to make good use of Fugen as a place for technological development which is opened domestic and international, as the central place in research and development base of Fukui prefecture, and to utilize the outcome in our decommissioning to the technical development effectively. This report consists of presentation paper are "Achievements and Considerations for Sampling and Analysis of Reactor Core Components", "Treatment of liquid scintillator waste liquid" and "Results and issues of rationalization of decontamination related to the clearance and considerations related to surface contamination monitoring" which is presented in the 39th Technical Special Committee on Fugen Decommissioning.
Nakanishi, Takumi*; Hori, Yuta*; Shigeta, Yasuteru*; Sato, Hiroyasu*; Kiyanagi, Ryoji; Munakata, Koji*; Ohara, Takashi; Okazawa, Atsushi*; Shimada, Rintaro*; Sakamoto, Akira*; et al.
Journal of the American Chemical Society, 145(35), p.19177 - 19181, 2023/08
Times Cited Count:0 Percentile:0(Chemistry, Multidisciplinary)Hayashi, Hirokazu; Tsubata, Yasuhiro; Sato, Takumi
Nihon Genshiryoku Gakkai Wabun Rombunshi (Internet), 22(3), p.97 - 107, 2023/08
The Japan Atomic Energy Agency has chosen nitride fuel as the first candidate for the transmutation of long-lived minor actinides (MA) using accelerator-driven systems (ADS). The pyrochemical method has been considered for reprocessing spent MA nitride fuels, because their decay heat should be very large for aqueous reprocessing. This study was conducted to investigate the effect of decay heat on the pyrochemical reprocessing of MA nitride fuels. On the basis of the estimated decay heats and the temperature limits of the materials that are to be handled in pyrochemical reprocessing, quantities adequate for handling in argon gas atmosphere were evaluated. From these considerations, we proposed that an electrorefiner with a diameter of 26 cm comprising 12 cadmium (Cd) cathodes with a diameter of 4 cm is suitable. On the basis of the size of the electrorefiner, the number necessary to reprocess spent MA fuels from 1 ADS in 200 days was evaluated to be 25. Furthermore, the amount of Cd-actinides (An) alloy to produce An nitrides by the nitridation-distillation combined reaction process was proposed to be about one-quarter that of Cd-An cathode material. The evaluated sizes and required numbers of equipment support the feasibility of pyrochemical reprocessing for MA nitride fuels.
 and Zr at precisely controlled high temperatures
and Zr at precisely controlled high temperaturesShirasu, Noriko; Sato, Takumi; Suzuki, Akihiro*; Nagae, Yuji; Kurata, Masaki
Journal of Nuclear Science and Technology, 60(6), p.697 - 714, 2023/06
Times Cited Count:1 Percentile:72.91(Nuclear Science & Technology)Interaction tests between UO and Zr were performed at precisely controlled high temperatures between 1840 and 2000
and Zr were performed at precisely controlled high temperatures between 1840 and 2000  C to understand the interaction mechanism in detail. A Zr rod was inserted in a UO
C to understand the interaction mechanism in detail. A Zr rod was inserted in a UO crucible and then heat-treated at a fixed temperature in Ar-gas flow for 10 min. After heating in the range of 1890 to 1930
crucible and then heat-treated at a fixed temperature in Ar-gas flow for 10 min. After heating in the range of 1890 to 1930  C, the Zr rod was deformed to a round shape, in which the post-analysis detected the significant diffusion of U into the Zr region and the formation of a dominant
C, the Zr rod was deformed to a round shape, in which the post-analysis detected the significant diffusion of U into the Zr region and the formation of a dominant  -Zr(O) matrix and a small amount of U-Zr-O precipitates. The abrupt progress of liquefaction was observed in the sample heated at around 1940
-Zr(O) matrix and a small amount of U-Zr-O precipitates. The abrupt progress of liquefaction was observed in the sample heated at around 1940  C or higher. The higher oxygen concentration in the
C or higher. The higher oxygen concentration in the  -Zr(O) matrix suppressed the liquefaction progress, due to the variation in the equilibrium state. The U-Zr-O melt formation progressed by the selective dissolution of Zr from the matrix, and the selective diffusion of U could occur via the U-Zr-O melt.
-Zr(O) matrix suppressed the liquefaction progress, due to the variation in the equilibrium state. The U-Zr-O melt formation progressed by the selective dissolution of Zr from the matrix, and the selective diffusion of U could occur via the U-Zr-O melt.
Nakanishi, Takumi*; Hori, Yuta*; Shigeta, Yasuteru*; Sato, Hiroyasu*; Wu, S.-Q.*; Kiyanagi, Ryoji; Munakata, Koji*; Ohara, Takashi; Sato, Osamu*
Physical Chemistry Chemical Physics, 25(17), p.12394 - 12400, 2023/05
Times Cited Count:1 Percentile:56.86(Chemistry, Physical) -edge for Ce in bauxites using transition-edge sensors; Implications for Ti-rich geological samples
-edge for Ce in bauxites using transition-edge sensors; Implications for Ti-rich geological samplesLi, W.*; Yamada, Shinya*; Hashimoto, Tadashi; Okumura, Takuma*; Hayakawa, Ryota*; Nitta, Kiyofumi*; Sekizawa, Oki*; Suga, Hiroki*; Uruga, Tomoya*; Ichinohe, Yuto*; et al.
Analytica Chimica Acta, 1240, p.340755_1 - 340755_9, 2023/02
Times Cited Count:2 Percentile:31.9(Chemistry, Analytical)no abstracts in English
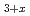 (M = Gd, Np) by the reaction of MN with Pd and chlorination of MPd
(M = Gd, Np) by the reaction of MN with Pd and chlorination of MPd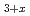 using cadmium chloride
using cadmium chlorideHayashi, Hirokazu; Shibata, Hiroki; Sato, Takumi; Otobe, Haruyoshi
Journal of Radioanalytical and Nuclear Chemistry, 332(2), p.503 - 510, 2023/02
Times Cited Count:0 Percentile:0.01(Chemistry, Analytical)The formation of MPd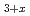 (M = Gd, Np) by the reaction of MN with Pd at 1323 K in Ar gas flow was observed. Cubic AuCu
(M = Gd, Np) by the reaction of MN with Pd at 1323 K in Ar gas flow was observed. Cubic AuCu -type GdPd
-type GdPd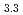 (
(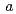 = 0.4081
= 0.4081  0.0001 nm) and NpPd
0.0001 nm) and NpPd (
(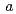 = 0.4081
= 0.4081  0.0001 nm) were identified, respectively. The product obtained from the reaction of NpN with Pd contained additional phases including the hexagonal TiNi
0.0001 nm) were identified, respectively. The product obtained from the reaction of NpN with Pd contained additional phases including the hexagonal TiNi -type NpPd
-type NpPd . Chlorination of the MPd
. Chlorination of the MPd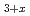 (M = Gd, Np) samples was accomplished by the solid-state reaction using cadmium chloride at 673 K in a dynamic vacuum. Pd-rich solid solution phase saturated with Cd and an intermetallic compound PdCd were obtained as by-products of MCl
(M = Gd, Np) samples was accomplished by the solid-state reaction using cadmium chloride at 673 K in a dynamic vacuum. Pd-rich solid solution phase saturated with Cd and an intermetallic compound PdCd were obtained as by-products of MCl formation.
formation.
Sato, Takumi; Nagae, Yuji; Kurata, Masaki; Quaini, A.*; Gu neau, C.*
neau, C.*
CALPHAD; Computer Coupling of Phase Diagrams and Thermochemistry, 79, p.102481_1 - 102481_11, 2022/12
Times Cited Count:0 Percentile:0.01(Thermodynamics)Xu, S.*; Odaira, Takumi*; Sato, Shunsuke*; Xu, X.*; Omori, Toshihiro*; Harjo, S.; Kawasaki, Takuro; Seiner, H.*; Zoubkov , K.*; Murakami, Yasukazu*; et al.
, K.*; Murakami, Yasukazu*; et al.
Nature Communications (Internet), 13, p.5307_1 - 5307_8, 2022/09
Times Cited Count:8 Percentile:66.14(Multidisciplinary Sciences)Yamashita, Takuya; Sato, Takumi; Madokoro, Hiroshi; Nagae, Yuji
Annals of Nuclear Energy, 173, p.109129_1 - 109129_15, 2022/08
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology) O
O -pentadentate planar ligands designed for the strongest and selective capture of uranium from seawater
-pentadentate planar ligands designed for the strongest and selective capture of uranium from seawaterMizumachi, Takumi*; Sato, Minami*; Kaneko, Masashi; Takeyama, Tomoyuki*; Tsushima, Satoru*; Takao, Koichiro*
Inorganic Chemistry, 61(16), p.6175 - 6181, 2022/04
Times Cited Count:2 Percentile:36.89(Chemistry, Inorganic & Nuclear)Based on unique 5-fold equatorial coordination of UO
 , water-compatible pentadentate planar ligands, H
, water-compatible pentadentate planar ligands, H saldian and its derivatives, were designed as strong and selective capture of UO
saldian and its derivatives, were designed as strong and selective capture of UO
 in seawater. In the simulated seawater condition (0.5 M NaCl + 2.3 mM HCO
in seawater. In the simulated seawater condition (0.5 M NaCl + 2.3 mM HCO
 /CO
/CO
 , pH 8), saldian
, pH 8), saldian shows the strongest complexation with UO
shows the strongest complexation with UO
 to form UO
to form UO (saldian) (log
(saldian) (log
 = 28.05
= 28.05  0.07), which is more than 10 order of magnitude greater than amidoxime-based or -inspired ligand systems most commonly employed for U capture from seawater. Good selectivity for UO
0.07), which is more than 10 order of magnitude greater than amidoxime-based or -inspired ligand systems most commonly employed for U capture from seawater. Good selectivity for UO
 from other metal ions coexisting in seawater was also demonstrated.
from other metal ions coexisting in seawater was also demonstrated.
Morishita, Kazuki; Sato, Takumi; Onishi, Takashi; Seki, Takayuki*; Sekine, Shinichi*; Okitsu, Yuichi*
JAEA-Technology 2021-024, 27 Pages, 2021/10
In the case of Plutonium (Pu)-bearing organic materials, organic materials are decomposed by alpha rays emitted mainly from Pu to generate hydrogen gas and other substances. Therefore, to safely store Pu-bearing organic materials for an extended period of time, organic materials must be eliminated. In addition, carbide and nitride fuels must be converted into oxides for safe storage in order to prevent the exothermal reaction of these fuels with oxygen/moisture in air. A survey of the literature on the stabilization treatment of Pu-bearing organic materials confirmed that organic materials can be decomposed and removed by heating at 950  C (1223.15 K) or greater in air. Furthermore, based on the calculated thermodynamic parameters of oxidation reaction of carbide and nitride fuels in air, it was estimated that these fuels would be oxidized in air at 950
C (1223.15 K) or greater in air. Furthermore, based on the calculated thermodynamic parameters of oxidation reaction of carbide and nitride fuels in air, it was estimated that these fuels would be oxidized in air at 950  C because the equilibrium oxygen partial pressure in the oxidation reaction at 950
C because the equilibrium oxygen partial pressure in the oxidation reaction at 950  C was lower than 2.1
C was lower than 2.1 10
10 Pa (oxygen partial pressure in air). Therefore, it was decided to stabilize Pu-bearing organic materials by heating at 950
Pa (oxygen partial pressure in air). Therefore, it was decided to stabilize Pu-bearing organic materials by heating at 950  C in air to remove the organic materials and oxidize the carbide and nitride fuels. As a mock-up test to remove the organic materials, thin sheets of epoxy resin were heated in air. The changes in appearance and weight before and after heating in air showed that organic materials can be removed. After the mock-up test, Pu-bearing organic materials were also stabilized by heating in the similar condition.
C in air to remove the organic materials and oxidize the carbide and nitride fuels. As a mock-up test to remove the organic materials, thin sheets of epoxy resin were heated in air. The changes in appearance and weight before and after heating in air showed that organic materials can be removed. After the mock-up test, Pu-bearing organic materials were also stabilized by heating in the similar condition.
 O
O ; Application of Raman imaging technique to uranium compound
; Application of Raman imaging technique to uranium compoundKusaka, Ryoji; Kumagai, Yuta; Yomogida, Takumi; Takano, Masahide; Watanabe, Masayuki; Sasaki, Takayuki*; Akiyama, Daisuke*; Sato, Nobuaki*; Kirishima, Akira*
Journal of Nuclear Science and Technology, 58(6), p.629 - 634, 2021/06
Times Cited Count:7 Percentile:66.68(Nuclear Science & Technology)Sudo, Ayako; Sato, Takumi; Ogi, Hiroshi; Takano, Masahide
Journal of Nuclear Science and Technology, 58(4), p.473 - 481, 2021/04
Times Cited Count:5 Percentile:65.59(Nuclear Science & Technology)Dissolution behavior of Sr and Ba is crucial for evaluating secondary source terms via coolant water from ex-vessel debris accumulated at Fukushima Daiichi Nuclear Power Plant. To understand the mechanism, knowing the distribution of Sr and Ba in the ex-vessel debris is necessary. As a result of reaction tests between simulated corium and concrete materials, two layered structures were observed in the solidified sample, (A) a silicate glass-based ((Si-Al-Ca-Fe-Zr-Cr-U-Sr-Ba)-O) phase-rich layer in the upper surface region and (B) a (U,Zr)O particle-rich layer at the inner region. Measurable concentrations of Sr and Ba were observed in layer (A) (approximately 1.7 times that in the layer (B)). According to thermodynamic analysis, (U,Zr)O
particle-rich layer at the inner region. Measurable concentrations of Sr and Ba were observed in layer (A) (approximately 1.7 times that in the layer (B)). According to thermodynamic analysis, (U,Zr)O is predicted to solidify, in advance, in the concrete-based melt around 2177
is predicted to solidify, in advance, in the concrete-based melt around 2177  C. Then, the residual melt is solidified as a silicate glass, and Sr and Ba are preferentially dissolved into the silicate glass. During the tests, (U,Zr)O
C. Then, the residual melt is solidified as a silicate glass, and Sr and Ba are preferentially dissolved into the silicate glass. During the tests, (U,Zr)O particles sank, in advance, in the melt because of its higher density, and the silicate glass phase relocated to the surface layer. On the other hand, silicate glass containing Sr and Ba is predicted to be hardly soluble in water and chemically stable.
particles sank, in advance, in the melt because of its higher density, and the silicate glass phase relocated to the surface layer. On the other hand, silicate glass containing Sr and Ba is predicted to be hardly soluble in water and chemically stable.
 Se
Se ultrathin films on van der Waals ferromagnet Cr
ultrathin films on van der Waals ferromagnet Cr Si
Si Te
Te
Kato, Takemi*; Sugawara, Katsuaki*; Ito, Naohiro*; Yamauchi, Kunihiko*; Sato, Takumi*; Oguchi, Tamio*; Takahashi, Takashi*; Shiomi, Yuki*; Saito, Eiji; Sato, Takafumi*
Physical Review Materials (Internet), 4(8), p.084202_1 - 084202_6, 2020/08
Times Cited Count:4 Percentile:20.66(Materials Science, Multidisciplinary)