Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yoshida, Keisuke; Kato, Shingo; Okuyama, Shinichi; Ishimori, Yuu; Inoue, Mutsuo*
Journal of Nuclear and Radiochemical Sciences (Internet), 24, p.1 - 12, 2024/08
 He cryostat and a closed-cycle
He cryostat and a closed-cycle  He gas handling system installed in a SQUID magnetometer without continuous-cooling functionality
He gas handling system installed in a SQUID magnetometer without continuous-cooling functionalityShimamura, Kazutoshi*; Wajima, Hiroki*; Makino, Hayato*; Abe, Satoshi*; Haga, Yoshinori; Sato, Yoshiaki*; Kawae, Tatsuya*; Yoshida, Yasuo*
Japanese Journal of Applied Physics, 61(5), p.056502_1 - 056502_7, 2022/05
Times Cited Count:3 Percentile:24.69(Physics, Applied)Mihara, Morihiro; Harasawa, Shuichi*; Torii, Kazuyuki*
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 26(1), p.15 - 23, 2019/06
Hardened cement pastes (HCPs) of water/cement ratio (W/C) 50% and 30% using fly ash (FA), blast furnace slag (BFS) and silica fume (SF) for 28 days were prepared. Apparent diffusion coefficients (D ) of Cs and I in HCPs were obtained using electron probe microanalysis. For Cs, BFS and SF contributed to a reduction of D
) of Cs and I in HCPs were obtained using electron probe microanalysis. For Cs, BFS and SF contributed to a reduction of D for W/C=50% and 30%, respectively. For I, BFS reduced D
for W/C=50% and 30%, respectively. For I, BFS reduced D for W/C=50%, however a significant reduction in D
for W/C=50%, however a significant reduction in D for W/C=30% was not observed. Using SF enhanced sorption of Cs on HCP, and using BFS slightly improved sorption of Cs on HCP. It was also confirmed that the pore structures of HCP using SF and BFS were connected by fine pores. It was therefore considered that using SF and BFS contributes to the reduction of D
for W/C=30% was not observed. Using SF enhanced sorption of Cs on HCP, and using BFS slightly improved sorption of Cs on HCP. It was also confirmed that the pore structures of HCP using SF and BFS were connected by fine pores. It was therefore considered that using SF and BFS contributes to the reduction of D in HCP.
in HCP.
 acidic solutions
acidic solutionsYokoyama, Akihiko*; Kitayama, Yuta*; Fukuda, Yoshiki*; Kikunaga, Hidetoshi*; Murakami, Masashi*; Komori, Yukiko*; Yano, Shinya*; Haba, Hiromitsu*; Tsukada, Kazuaki; Toyoshima, Atsushi*
Radiochimica Acta, 107(1), p.27 - 32, 2019/01
Times Cited Count:1 Percentile:8.72(Chemistry, Inorganic & Nuclear)Koarashi, Jun; Nishimura, Shusaku; Atarashi-Andoh, Mariko; Matsunaga, Takeshi*; Sato, Tsutomu*; Nagao, Seiya*
Chemosphere, 205, p.147 - 155, 2018/08
Times Cited Count:21 Percentile:53.86(Environmental Sciences)There is little understanding of how soil aggregation can affect the mobility and bioavailability of  Cs in soils. To explore this, soil samples were collected at seven sites under different land-use conditions in Fukushima and were separated into four aggregate-size fractions. The fractions were then analyzed for
Cs in soils. To explore this, soil samples were collected at seven sites under different land-use conditions in Fukushima and were separated into four aggregate-size fractions. The fractions were then analyzed for  Cs content and extractability and mineral composition. In forest soils, aggregate formation was significant, and
Cs content and extractability and mineral composition. In forest soils, aggregate formation was significant, and  Cs was largely associated with large-sized aggregates. In contrast, there was less aggregation in agricultural field soils, and most of
Cs was largely associated with large-sized aggregates. In contrast, there was less aggregation in agricultural field soils, and most of  Cs was in the clay- and silt-sized fractions. Across all sites, the
Cs was in the clay- and silt-sized fractions. Across all sites, the  Cs extractability was higher in the large-sized aggregate fractions than in the clay-sized fractions. The results demonstrate that large-sized aggregates are a significant reservoir of potentially mobile and bioavailable
Cs extractability was higher in the large-sized aggregate fractions than in the clay-sized fractions. The results demonstrate that large-sized aggregates are a significant reservoir of potentially mobile and bioavailable  Cs in organic-rich (forest and orchard) soils.
Cs in organic-rich (forest and orchard) soils.
Hatakeyama, Mizuki*; Nishiyama, Yoshio*; Nagatani, Hirohisa*; Okamura, Hiroyuki; Imura, Hisanori*
Solvent Extraction Research and Development, Japan, 25(2), p.79 - 89, 2018/00
Times Cited Count:13 Percentile:39.98(Chemistry, Multidisciplinary)The synergistic extraction of trivalent lanthanide ions (Ln(III)) with benzoylacetone (Hba) and trioctyphosphine oxide (TOPO) in an ionic liquid (IL), 1-butyl-3-methyl-imidazolium bis(trifluoromethanesulfonyl)imide ([Bmim][Tf N]), has been investigated. The extractability of Ln(III) with Hba was significantly enhanced in the presence of TOPO. The composition and extraction constants of the extracted species for each Ln(III) were determined by 3-dimensional equilibrium analysis. It was found that all Ln(III) were extracted as cationic ternary complexes such as Ln(ba)
N]), has been investigated. The extractability of Ln(III) with Hba was significantly enhanced in the presence of TOPO. The composition and extraction constants of the extracted species for each Ln(III) were determined by 3-dimensional equilibrium analysis. It was found that all Ln(III) were extracted as cationic ternary complexes such as Ln(ba) (TOPO)
(TOPO)
 or Ln(ba)(TOPO)
or Ln(ba)(TOPO)
 with Hba and TOPO. Furthermore, the formation constants of the cationic ternary complexes in the IL indicate that Lu(ba)(TOPO)
with Hba and TOPO. Furthermore, the formation constants of the cationic ternary complexes in the IL indicate that Lu(ba)(TOPO)
 is the most stable complex in the IL.
is the most stable complex in the IL.
Okamura, Hiroyuki
Bunseki Kagaku, 66(7), p.531 - 532, 2017/07
This paper summarizes the author's doctoral thesis on analytical chemistry. In this study, the ionic liquid (IL) extraction of various metal(II, III) complexes with macrocyclic and anionic chelating ligands has been investigated to clarify the solvent effect of ILs and to demonstrate the specificity and superiority of the IL extraction systems over conventional ones. The evaluation of the extraction equilibrium of Eu(III) with 2-thenoyltrifluoroacetone (Htta) in 1-alkyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([C mim][Tf
mim][Tf N]) and time-resolved laser-induced fluorescence spectroscopy revealed the specific solute-solvent interactions between Eu(tta)
N]) and time-resolved laser-induced fluorescence spectroscopy revealed the specific solute-solvent interactions between Eu(tta) and [C
and [C mim][Tf
mim][Tf N]. The IL synergistic extraction with Htta and
N]. The IL synergistic extraction with Htta and 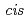 -dicyclohexano-18-crown-6 was developed. The extractability of lighter lanthanides(III) was remarkably enhanced by a synergistic effect of crown ethers. A macrocyclic ligand (H
-dicyclohexano-18-crown-6 was developed. The extractability of lighter lanthanides(III) was remarkably enhanced by a synergistic effect of crown ethers. A macrocyclic ligand (H
 DA18C6) composed of diaza-18-crown-6 and two 4-acyl-5-pyrazolones was synthesized for the extraction of Sr(II). The extraction performance of H
DA18C6) composed of diaza-18-crown-6 and two 4-acyl-5-pyrazolones was synthesized for the extraction of Sr(II). The extraction performance of H
 DA18C6 was significantly enhanced only in [C
DA18C6 was significantly enhanced only in [C mim][Tf
mim][Tf N] by the intramolecular cooperative effect.
N] by the intramolecular cooperative effect.
 N
N anions in the ionic liquid-water distribution of europium(III) chelates
anions in the ionic liquid-water distribution of europium(III) chelatesOkamura, Hiroyuki; Aoyagi, Noboru; Shimojo, Kojiro; Naganawa, Hirochika; Imura, Hisanori*
RSC Advances (Internet), 7(13), p.7610 - 7618, 2017/01
Times Cited Count:20 Percentile:52.59(Chemistry, Multidisciplinary)The role of bis(trifluoromethanesulfonyl)imide (Tf N
N ) anions in the ionic liquid-water distribution systems of the Eu(III) chelates with 2-thenoyltrifluoroacetone (Htta) was investigated by the liquid-liquid distribution and time-resolved laser-induced fluorescence spectroscopy (TRLFS). The effect of the ionic liquids on the distribution constant of Eu(tta)
) anions in the ionic liquid-water distribution systems of the Eu(III) chelates with 2-thenoyltrifluoroacetone (Htta) was investigated by the liquid-liquid distribution and time-resolved laser-induced fluorescence spectroscopy (TRLFS). The effect of the ionic liquids on the distribution constant of Eu(tta) was evaluated by the regular solution theory. The distribution constant of Eu(tta)
was evaluated by the regular solution theory. The distribution constant of Eu(tta) in 1-alkyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([C
in 1-alkyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([C mim][Tf
mim][Tf N]) was increased dramatically by the solvation effects of Eu(tta)
N]) was increased dramatically by the solvation effects of Eu(tta) in [C
in [C mim][Tf
mim][Tf N]. TRLFS for [Eu(tta)
N]. TRLFS for [Eu(tta) (H
(H O)
O) ] synthesized revealed that the Eu(tta)
] synthesized revealed that the Eu(tta) chelate was almost completely dehydrated in a series of [C
chelate was almost completely dehydrated in a series of [C mim][Tf
mim][Tf N]. The Eu(tta)
N]. The Eu(tta) chelate exists as di- or tri-hydrates in 1-ethyl-3-methylimidazolium perchlorate ([C
chelate exists as di- or tri-hydrates in 1-ethyl-3-methylimidazolium perchlorate ([C mim][ClO
mim][ClO ]) containing 20 M water, whereas mono-hydrated chelate was formed in [C
]) containing 20 M water, whereas mono-hydrated chelate was formed in [C mim][Tf
mim][Tf N, ClO
N, ClO ] in the presence of 0.50 M Tf
] in the presence of 0.50 M Tf N
N and 20 M water. These results show that the coordinated water molecules of [Eu(tta)
and 20 M water. These results show that the coordinated water molecules of [Eu(tta) (H
(H O)
O) ] were replaced by the Tf
] were replaced by the Tf N
N anions. In fact, an anionic adduct, [Eu(tta)
anions. In fact, an anionic adduct, [Eu(tta) (Tf
(Tf N)]
N)] , was observed by electrospray ionization mass spectrometry in the presence of [C
, was observed by electrospray ionization mass spectrometry in the presence of [C mim][Tf
mim][Tf N].
N].
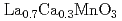
Sato, Wataru*; Komatsuda, Sayaka*; Osa, Akihiko; Sato, Tetsuya; Okubo, Yoshitaka*
Hyperfine Interactions, 237(1), p.113_1 - 113_6, 2016/12
Times Cited Count:2 Percentile:59.53(Physics, Atomic, Molecular & Chemical)The magnetic hyperfine field and electric field gradient at the Cd(
Cd(
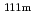 Cd) and
Cd) and 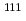 Cd(
Cd(
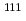 In) probe nuclei introduced in a perovskite manganese oxide
In) probe nuclei introduced in a perovskite manganese oxide 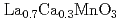 (
(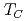
 250 K) were measured for the study of the local magnetism and structure by means of time-differential perturbed angular correlation spectroscopy. In the ferromagnetic phase at 77 K, a very slight supertransferred magnetic hyperfine field (SMHF) (
250 K) were measured for the study of the local magnetism and structure by means of time-differential perturbed angular correlation spectroscopy. In the ferromagnetic phase at 77 K, a very slight supertransferred magnetic hyperfine field (SMHF) ( 0.014 T) combined with a well-defined electric field gradient was observed at the nonmagnetic
0.014 T) combined with a well-defined electric field gradient was observed at the nonmagnetic  Cd nucleus on the La/Ca A site. This observation suggests that the large magnetic hyperfine field (
Cd nucleus on the La/Ca A site. This observation suggests that the large magnetic hyperfine field (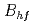 = 6.9 T) measured, in our previous work, at the
= 6.9 T) measured, in our previous work, at the 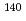 Ce probe nucleus on the A site originates from the contribution of a 4
Ce probe nucleus on the A site originates from the contribution of a 4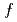 spin oriented by the SMHF from adjacent Mn ions.
spin oriented by the SMHF from adjacent Mn ions.
 Cs in a forested catchment affected by the Fukushima Daiichi Nuclear Power Plant accident
Cs in a forested catchment affected by the Fukushima Daiichi Nuclear Power Plant accidentMatsunaga, Takeshi; Nakanishi, Takahiro; Atarashi-Andoh, Mariko; Takeuchi, Erina; Muto, Kotomi; Tsuzuki, Katsunori; Nishimura, Shusaku; Koarashi, Jun; Otosaka, Shigeyoshi; Sato, Tsutomu*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 310(2), p.679 - 693, 2016/11
Times Cited Count:6 Percentile:45.07(Chemistry, Analytical)Particulate  Cs in stream water was collected continuously for two years in order to assess the long-term trend of the
Cs in stream water was collected continuously for two years in order to assess the long-term trend of the  Cs discharge from the forest environment. Sampling was conducted from December 2011 to December 2013 in a mountainous stream, which received the
Cs discharge from the forest environment. Sampling was conducted from December 2011 to December 2013 in a mountainous stream, which received the  Cs from the Fukushima Daiichi Nuclear Power Plant accident. A seasonal increase in fluvial transport load of particulate
Cs from the Fukushima Daiichi Nuclear Power Plant accident. A seasonal increase in fluvial transport load of particulate  Cs associated with suspended solids (SS) was observed in August and September when rainfall was abundant. The particulate
Cs associated with suspended solids (SS) was observed in August and September when rainfall was abundant. The particulate  Cs concentration decreased at a faster rate than the rate due to radioactive decay. This decrease might be resulted from redistribution of the easily eroded and polluted soil surface due to heavy rain events such as typhoons. These findings indicate that the particulate
Cs concentration decreased at a faster rate than the rate due to radioactive decay. This decrease might be resulted from redistribution of the easily eroded and polluted soil surface due to heavy rain events such as typhoons. These findings indicate that the particulate  Cs load was subject to the inter-annual variations in rainfalls, and decreased gradually over a long period of time due to a decrease in
Cs load was subject to the inter-annual variations in rainfalls, and decreased gradually over a long period of time due to a decrease in  Cs concentration in SS.
Cs concentration in SS.
 radionuclide therapy
radionuclide therapyOgawa, Kazuma*; Mizuno, Yoshiaki*; Washiyama, Koshin*; Shiba, Kazuhiro*; Takahashi, Naruto*; Kozaka, Takashi*; Watanabe, Shigeki; Shinohara, Atsushi*; Odani, Akira*
Nuclear Medicine and Biology, 42(11), p.875 - 879, 2015/11
Times Cited Count:27 Percentile:72.67(Radiology, Nuclear Medicine & Medical Imaging)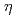
 -semiquinone complex; A Possible dielectric response mechanism
-semiquinone complex; A Possible dielectric response mechanismMitsumi, Minoru*; Ezaki, Kazunari*; Komatsu, Yuki*; Toriumi, Koshiro*; Miyato, Tatsuya*; Mizuno, Motohiro*; Azuma, Nobuaki*; Miyazaki, Yuji*; Nakano, Motohiro*; Kitagawa, Yasutaka*; et al.
Chemistry; A European Journal, 21(27), p.9682 - 9696, 2015/06
Times Cited Count:10 Percentile:31.10(Chemistry, Multidisciplinary)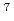 Li +
Li + 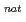 Pb reaction
Pb reactionNishinaka, Ichiro; Yokoyama, Akihiko*; Washiyama, Koshin*; Maeda, Eita*; Watanabe, Shigeki; Hashimoto, Kazuyuki; Ishioka, Noriko; Makii, Hiroyuki; Toyoshima, Atsushi; Yamada, Norihiro*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 304(3), p.1077 - 1083, 2015/06
Times Cited Count:10 Percentile:60.46(Chemistry, Analytical)Production cross sections of astatine isotopes 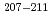 At in the 29-57 MeV
At in the 29-57 MeV 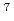 Li induced reaction with
Li induced reaction with 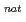 Pb target have been measured by
Pb target have been measured by  - and
- and  -ray spectrometry. Excitation functions of production cross sections have been compared with a statistical model calculation to study the reaction mechanism of
-ray spectrometry. Excitation functions of production cross sections have been compared with a statistical model calculation to study the reaction mechanism of 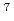 Li +
Li +  Pb. Considerably small experimental cross sections of
Pb. Considerably small experimental cross sections of 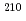 At and
At and 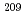 At compared with the calculation were clearly observed at incident energies higher than 44 MeV, indicating that the effects of breakup reaction play a role. A chemical separation of astatine from an irradiated lead target has been studied with a dry-distillation method. A complementary way to produce astatine isotopes has been developed.
At compared with the calculation were clearly observed at incident energies higher than 44 MeV, indicating that the effects of breakup reaction play a role. A chemical separation of astatine from an irradiated lead target has been studied with a dry-distillation method. A complementary way to produce astatine isotopes has been developed.
Oe, Kazuhiro*; Attallah, M. F.*; Asai, Masato; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke*; Kasamatsu, Yoshitaka*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1317 - 1320, 2015/02
Times Cited Count:10 Percentile:60.46(Chemistry, Analytical)A new technique for continuous dissolution of nuclear reaction products transported by a gas-jet system was developed for superheavy element (SHE) chemistry. In this technique, a hydrophobic membrane is utilized to separate an aqueous phase from the gas phase. With this technique, the dissolution efficiencies of short-lived radionuclides of 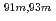 Mo and
Mo and 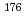 W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
 Rn-
Rn- At generator
At generatorMaeda, Eita*; Yokoyama, Akihiko*; Taniguchi, Takumi*; Washiyama, Koshin*; Nishinaka, Ichiro
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1465 - 1468, 2015/02
Times Cited Count:12 Percentile:67.00(Chemistry, Analytical)The  At isotope has gathered attention as a promising
At isotope has gathered attention as a promising  -emitter for radionuclide therapy. We report the dependence of the distribution ratio of astatine on the concentration of HCl, and on the polarity of the organic solvent. The results will be useful for development of the
-emitter for radionuclide therapy. We report the dependence of the distribution ratio of astatine on the concentration of HCl, and on the polarity of the organic solvent. The results will be useful for development of the  Rn-
Rn- At generator.
At generator.
Matsunaga, Takeshi; Nakanishi, Takahiro; Atarashi-Andoh, Mariko; Takeuchi, Erina; Tsuzuki, Katsunori; Nishimura, Shusaku; Koarashi, Jun; Otosaka, Shigeyoshi; Sato, Tsutomu*; Nagao, Seiya*
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1291 - 1295, 2015/02
Times Cited Count:3 Percentile:23.43(Chemistry, Analytical)An innovative, yet simple method for the passive collection of radioactive materials in river water has been developed and validated. The method employes large filter vessels, containing multiple cartridge filters. River water is led to the system naturally using a drop of the riverbed by hose from upstream. This method makes long-term, unmanned monitoring possible. In addition to regular radioactivity analyses, this method provides an opportunity for the characterization of suspended materials based on its ample collection quantities (more than several tens of grams). This method may also be applicable to sediment-bound chemicals.
 -diketone fragments on the extraction of strontium into an ionic liquid
-diketone fragments on the extraction of strontium into an ionic liquidOkamura, Hiroyuki; Naganawa, Hirochika; Imura, Hisanori*; Shimojo, Kojiro
Proceedings of 20th International Solvent Extraction Conference (ISEC 2014), p.1046 - 1051, 2014/09
 -diketones and trioctylphosphine oxide in an ionic liquid
-diketones and trioctylphosphine oxide in an ionic liquidOkamura, Hiroyuki; Takagi, Hitomi*; Isomura, Taku*; Morita, Kotaro*; Nagatani, Hirohisa*; Imura, Hisanori*
Analytical Sciences, 30(3), p.323 - 325, 2014/03
Times Cited Count:32 Percentile:72.78(Chemistry, Analytical)Ninomiya, Kazuaki*; Omote, Sayuri*; Sato, Katsuya; Narumi, Issey*; Shimizu, Nobuaki*
JAEA-Review 2013-059, JAEA Takasaki Annual Report 2012, P. 115, 2014/03
Fukushi, Keisuke*; Hasegawa, Yusuke*; Maeda, Koshi*; Aoi, Yusuke*; Tamura, Akihiro*; Arai, Shoji*; Yamamoto, Yuhei*; Aosai, Daisuke*; Mizuno, Takashi
Environmental Science & Technology, 47(22), p.12811 - 12818, 2013/11
Times Cited Count:34 Percentile:62.33(Engineering, Environmental)Eu(III) sorption on granite was examined by the combined microscopic and macroscopic approaches. Polished thin sections of the granite were reacted with solutions containing 10  M of Eu(III) and analyzed using EPMA and LA-ICP-MS. The Eu enrichment up to 6 wt.% was observed on most of the biotite grains. The Eu-enriched parts commonly lose K, which is the interlayer cation of biotite, indicating that the sorption mode is cation exchange in the interlayer. Batch Eu(III) sorption experiments on granite and biotite powders were conducted. The macroscopic sorption behavior of biotite was consistent with that of granite. The obtained sorption edges can be reproduced reasonably by the modeling considering single-site cation exchange reactions. Granite is complex mineral assemblages. However, the combined microscopic and macroscopic approaches revealed that elementary reactions by single phase can be representative for the bulk sorption reaction in complex mineral assemblages.
M of Eu(III) and analyzed using EPMA and LA-ICP-MS. The Eu enrichment up to 6 wt.% was observed on most of the biotite grains. The Eu-enriched parts commonly lose K, which is the interlayer cation of biotite, indicating that the sorption mode is cation exchange in the interlayer. Batch Eu(III) sorption experiments on granite and biotite powders were conducted. The macroscopic sorption behavior of biotite was consistent with that of granite. The obtained sorption edges can be reproduced reasonably by the modeling considering single-site cation exchange reactions. Granite is complex mineral assemblages. However, the combined microscopic and macroscopic approaches revealed that elementary reactions by single phase can be representative for the bulk sorption reaction in complex mineral assemblages.