Crystal structure of UDP-glucose: anthocyanidin 3- - glucosyltransferase from
- glucosyltransferase from 
チョウマメ花弁由来UDP-グルコース: アントシアニジン3- -グルコシルトランスフェラーゼのX線結晶構造解析
-グルコシルトランスフェラーゼのX線結晶構造解析
廣本 武史; 本庄 栄二郎*; 玉田 太郎; 黒木 良太
Hiromoto, Takeshi; Honjo, Eijiro*; Tamada, Taro; Kuroki, Ryota
UDP-glucose: anthocyanidin 3- -glucosyltransferase from
-glucosyltransferase from  (
( 3GT-A; AB185904) is an enzyme that catalyses glucosyl transfer from UDP-glucose to anthocyanidins such as delphinidin, which is the first step of ternatin biosynthesis (Kogawa
3GT-A; AB185904) is an enzyme that catalyses glucosyl transfer from UDP-glucose to anthocyanidins such as delphinidin, which is the first step of ternatin biosynthesis (Kogawa  ., 2007). The recombinant wild-type enzyme was expressed in
., 2007). The recombinant wild-type enzyme was expressed in  cells, purified to homogeneity and crystallized. The X-ray diffraction data set was collected at PF-BL6A using a crystal with a size of 0.05
cells, purified to homogeneity and crystallized. The X-ray diffraction data set was collected at PF-BL6A using a crystal with a size of 0.05  0.05
0.05  0.5 mm, which belongs to the space group
0.5 mm, which belongs to the space group 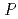 2
2 with cell dimensions of
with cell dimensions of 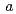 = 50.2
= 50.2  ,
, 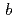 = 55.2
= 55.2  ,
,  = 86.2
= 86.2  and
and  = 105.1
= 105.1  . The initial phase was solved by MR using the coordinates of a homologous glucosyltransferase
. The initial phase was solved by MR using the coordinates of a homologous glucosyltransferase 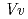 GT1 from
GT1 from 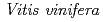 (PDB ID: 2C1Z) as a search model. The overall structure of
(PDB ID: 2C1Z) as a search model. The overall structure of  3GT-A shows a typical GT-B fold comprising two Rossmann-like
3GT-A shows a typical GT-B fold comprising two Rossmann-like  /
/ /
/ domains. The putative binding sites for UDP-glucose and delphinidin are located in a deep cleft between the N- and C-terminal domains. Structural homology searches by Dali server indicated that
domains. The putative binding sites for UDP-glucose and delphinidin are located in a deep cleft between the N- and C-terminal domains. Structural homology searches by Dali server indicated that  3GT-A is similar to the other plant glucosyltransferases
3GT-A is similar to the other plant glucosyltransferases 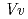 GT1 and UGT78G1 from
GT1 and UGT78G1 from 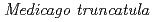 with the RMS deviations of 1.9
with the RMS deviations of 1.9  (for 432 C
(for 432 C atoms) and 2.0
atoms) and 2.0  (for 437 C
(for 437 C atoms), respectively.
atoms), respectively.