Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Taniguchi, Naoki; Kawasaki, Manabu*; Fujiwara, Kazuo*
JNC TN8400 2001-011, 62 Pages, 2001/03
The corrosion of metallic materials used in natural environment are sometimes affected by microbial action. It is apprehended that microorganism living in deep underground or brought from ground surface during excavation makes an impact on overpack material for geological disposal of high-level radioactive waste. Sulfate reducing bacteria (SRB) is known to be one of the most representative microorganism which affects the corrosion of metals. In this study, the behavior of growth of SRB was investigated at first under the presence of bentonite as a main component of buffer material which encloses the overpack. The results of the tests showed that the population of SRB after the culture in synthetic sea water mixed with bentonite decreased with increasing the ratio of bentonite/solution. SRB was hardly grown in medium whose bentonite/solution ratio exceeded 1000g/l. As a conservative case, the effects of sulfide on the corrosion of overpack materials were also studied assuming high activity of SRB. Carbon steel, copper and titanium specimens were immersed in synthetic sea water purging 0.1MPa H S gas and the corrosion behavior was compared with the results in N
S gas and the corrosion behavior was compared with the results in N gas purging environment. Obvious effect of sulfide on the corrosion of carbon steel was not observed, but the corrosion rates of copper specimens were accelerated several hundred times by purging H
gas purging environment. Obvious effect of sulfide on the corrosion of carbon steel was not observed, but the corrosion rates of copper specimens were accelerated several hundred times by purging H S gas. The absorption of hydrogen into titanium specimens was not affected by purging H
S gas. The absorption of hydrogen into titanium specimens was not affected by purging H S gas, but the difference of hydrogen absorption between pure titanium and titanium alloy containing 0.06%-Pd was observed.
S gas, but the difference of hydrogen absorption between pure titanium and titanium alloy containing 0.06%-Pd was observed.
Iwase, Masanori*
JNC TJ8400 2000-063, 78 Pages, 2000/03
This study is aimed at controlling oxidation reaction of molten metal by ash in incineration systems, and at positively utilizing the oxidation reaction for decontamination of slag. In this year, in order to investigate physico-chemical properties of mixed fused salt containing alkali sulfates, with special focus on the behaviour of oxygen anion in the melts, Cu / Cu
/ Cu redox equilibrium experiments were carried out. Among the effect of various parameters on Cu
redox equilibrium experiments were carried out. Among the effect of various parameters on Cu / Cu
/ Cu ratio in binary and ternary alkali sulfate melts, the effect of partial pressures of oxygen and SO
ratio in binary and ternary alkali sulfate melts, the effect of partial pressures of oxygen and SO was mainly investigated in the study. Variation in Cu
was mainly investigated in the study. Variation in Cu / Cu
/ Cu ratio were presented as the function of partial pressures of oxygen and SO
ratio were presented as the function of partial pressures of oxygen and SO , respectively. Possible thermodynamic interpretation were made on the experimental results. In addition, the dissolution of Cr
, respectively. Possible thermodynamic interpretation were made on the experimental results. In addition, the dissolution of Cr O
O in mixed alkali sulfates were also investigated as a first step to elucidate the mechanism of hot corrosion. With this investigation, an important finding was obtained that the solubility of Cr
in mixed alkali sulfates were also investigated as a first step to elucidate the mechanism of hot corrosion. With this investigation, an important finding was obtained that the solubility of Cr O
O for melts with same average ionic radius, in other words, oxygen ion activity, were essentially identical under constant temperature and atmosphere.
for melts with same average ionic radius, in other words, oxygen ion activity, were essentially identical under constant temperature and atmosphere.
Oda, Chie; *
JNC TN8400 98-001, 14 Pages, 1998/11
Solubilities of amorphous stannic oxide, SnO (am) in Na-ClO
(am) in Na-ClO -Cl-SO
-Cl-SO aqueous systems were measured to quantitatively investigate the influences of the ligands OH
aqueous systems were measured to quantitatively investigate the influences of the ligands OH , Cl
, Cl and SO
and SO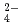 on solubilities. They were also measured in bentonite equilibrated solutions to discuss the behavior of tin under a repository condition of a high-revel radioactive waste. The solubility data in sodium perchlorate media in the range of pH from 6 to 11 showed pH dependency, and the hydrolysis constants of tin (IV) were determined (Amaya, et al., 1997). No significant changes in solubilities with the variation in Cl
on solubilities. They were also measured in bentonite equilibrated solutions to discuss the behavior of tin under a repository condition of a high-revel radioactive waste. The solubility data in sodium perchlorate media in the range of pH from 6 to 11 showed pH dependency, and the hydrolysis constants of tin (IV) were determined (Amaya, et al., 1997). No significant changes in solubilities with the variation in Cl , SO
, SO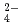 concentrations were observed in Na-ClO
concentrations were observed in Na-ClO -Cl-SO
-Cl-SO aqueous systems, so this indicates that chloride and sulfate species were less effective than hydroxide complexes. On the other hand, solubilities in bentonite equilibrated solutions were higher than solubilities of other experiments in simple systems. These results suggest that (1) other complexes of tin except hydroxide, chloride or sulfate complexes of tin (IV) may dominantly exist in aqueous phase, (2)solid phase other than SnO
aqueous systems, so this indicates that chloride and sulfate species were less effective than hydroxide complexes. On the other hand, solubilities in bentonite equilibrated solutions were higher than solubilities of other experiments in simple systems. These results suggest that (1) other complexes of tin except hydroxide, chloride or sulfate complexes of tin (IV) may dominantly exist in aqueous phase, (2)solid phase other than SnO (am) may limit the solubility of tin under repository conditions.
(am) may limit the solubility of tin under repository conditions.
Okoshi, Minoru
JAERI-Review 97-009, 32 Pages, 1997/08
no abstracts in English
 microspheres
microspheres; Takahashi, Yoshihisa
Journal of Nuclear Materials, 189, p.134 - 140, 1992/00
Times Cited Count:3 Percentile:35.29(Materials Science, Multidisciplinary)no abstracts in English
 microspheres
microspheres; Takahashi, Yoshihisa
Journal of Nuclear Materials, 182, p.195 - 202, 1991/00
Times Cited Count:5 Percentile:53.84(Materials Science, Multidisciplinary)no abstracts in English
; *;
Shikizai Kyokai-Shi, 56(7), p.443 - 448, 1983/00
no abstracts in English
Tagawa, Hiroaki
JAERI-M 6421, 34 Pages, 1976/03
no abstracts in English


 I and sulfite-
I and sulfite-
 S
S; ;
Journal of Nuclear Science and Technology, 13(3), p.125 - 131, 1976/03
Times Cited Count:4no abstracts in English

 S-labeled solid sulfite and thiosulfate aqueous solution
S-labeled solid sulfite and thiosulfate aqueous solution;
Radioisotopes, 25(1), p.39 - 41, 1976/01
no abstracts in English