Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 C
CMaeda, Toshikatsu; Chiba, Noriaki; Tateishi, Tsuyoshi*; Yamaguchi, Tetsuji
Nihon Genshiryoku Gakkai Wabun Rombunshi, 12(2), p.158 - 164, 2013/06
no abstracts in English
Otsuka, Ichiro*; Iida, Yoshihisa; Yamaguchi, Tetsuji; Kato, Osamu*; Tateishi, Tsuyoshi*; Tanaka, Tadao
Materials Research Society Symposium Proceedings, Vol.1475, p.507 - 512, 2012/06
We conducted corrosion experiments of carbon wires to identify the cathode reaction with overpack corrosion. Carbon steel wires were immersed in selected aqueous solutions of distilled water, sodium hydrogen carbonate solution and sodium sulfate solution sealed in glass ampoules. The ampoules were kept at 60  C for 150 days. Corrosion products, liquid phase and gas phases were analyzed. The results showed that hydrogen gas generation by reduction of hydrogen ions was dominant cathode reaction. Reduction of sulfate and carbonate anions was not observed. Amorphous ferrous hydroxide and/or iron oxides were identified in all the experiments as the corrosion products. Iron carbonate was also identified in the sodium hydrogen carbonate solution. This result indicates that the hydrogen generation reaction might be the dominant cathode reaction under geological disposal environment and that the Eh of the pore water would reach the equilibrium potential of the hydrogen generation reaction.
C for 150 days. Corrosion products, liquid phase and gas phases were analyzed. The results showed that hydrogen gas generation by reduction of hydrogen ions was dominant cathode reaction. Reduction of sulfate and carbonate anions was not observed. Amorphous ferrous hydroxide and/or iron oxides were identified in all the experiments as the corrosion products. Iron carbonate was also identified in the sodium hydrogen carbonate solution. This result indicates that the hydrogen generation reaction might be the dominant cathode reaction under geological disposal environment and that the Eh of the pore water would reach the equilibrium potential of the hydrogen generation reaction.
Honda, Akira; Masuda, Kaoru*; Tateishi, Tsuyoshi*; Kato, Osamu*; Inoue, Hiroyuki*
Zairyo To Kankyo, 60(12), p.541 - 552, 2011/12
Immersion tests and rest potential measurements under hyper-alkaline and high sodium nitrate concentrations were conducted to elucidate and improve model predictions of chemical interactions between carbon steel and nitrate ion in high concentrations of nitrate salt. The modified model can estimate the tendency of time dependent variation of chemical species and of rest potentials.
Taniguchi, Naoki; Suzuki, Hiroyuki*; Nakanishi, Tomoaki*; Nakayama, Takenori*; Masugata, Tsuyoshi*; Tateishi, Tsuyoshi*
Zairyo To Kankyo, 56(12), p.576 - 584, 2007/12
The long term hydrogen absorption behavior and the possibility of hydrogen embrittlement were studied for titanium overpack for high level radioactive waste disposal. The results of galvanostatic cathodic polarization tests showed that as the cathodic current density is lowered, the amount of absorbed hydrogen for a constant cathodic charge was increased as well as hydrogen permeated into inside of titanium. The hydrogen absorption ratio for a cathodic current density equivalent to the corrosion rate under anaerobic condition was estimated to nearly 100 percent, and the amount of absorbed hydrogen for 1000 years was evaluated to be 400 ppm. The mechanical property of titanium containing hydrogen depended on not only hydrogen concentration but also hydrogen distribution type. The more hydrogen distribution is uniform, the degree of embrittlement was larger. It was expected that the rupture of titanium overpack with 6 mm thickness would be initiated if the crack size in titanium is over about 2-3 mm under the stress corresponds to yield strength.
Honda, Akira; Kato, Takashi; Tateishi, Tsuyoshi*; Imakita, Tsuyoshi*; Masuda, Kaoru*; Kato, Osamu*; Nishimura, Tsutomu*
Zairyo To Kankyo, 55(10), p.458 - 465, 2006/10
Migration of radioactive material can be affected by the redox condition and the concentration of ligands in the repository of radioactive waste. It is possible that radioactive waste contains nitrate which can affect the migration behavior of radioactive nuclides by both changing the redox condition of the environment and acting as a ligand. On the other hand, several researchers observed the reduction of nitrate ions in ammonia due to the iron. Ammonia has a potential to ligand for radioactive nuclides. Nitrate can also affect the rate of hydrogen gas evolution accompanied by metal corrosion through changing the rest potential of metal by its oxidizing nature. Carbon steel was, therefore, immersed in an aqueous solution of sodium nitrate in a closed system for observing both the chemical interaction between metal and nitrate, and the effect of nitrate on the hydrogen gas evolution rate. The experimental pH range of the solution was 10.0-13.5 which corresponds to the pH range of pore fluid of cementitious material. The cathodic current density shows a Tafel equation type potential dependency in the aqueous solution containing nitrate or nitrite. In spite of the acceleration of cathodic reaction due to the existence of nitrate, the corrosion rates of carbon steel were not accelerated in the nitrate solutions. This fact suggests that the system is controlled by the anodic reaction. The nitrate reduction accompanied by the corrosion of carbon steel is considered to be a series reaction such as nitrate nitrite ammonia. The nitrate reduction reaction compete with the water reduction reaction within the anodic controlled condition, therefore nitrate strongly reduced the hydrogen evolution rate. The generation rates of ammonia were independent of the concentration of nitrate.
Honda, Akira; Masuda, Kaoru*; Kato, Osamu*; Nishimura, Tsutomu*; Tateishi, Tsuyoshi*; Imakita, Tsuyoshi*
JNC TN8400 2005-023, 40 Pages, 2005/09
Uranium and Plutonium are planed to be recovered from spent fuel by the reprocessing in Japan. PUREX method is internationally dominant among the commercial reprocessing plants. PUREX method has been also employed in Japan. The low level liquid waste from PUREX process would contain NO3- as forms of soluble salts, if the special process for decomposing NO3- were not adopted. The nitrate is possibly brought within the repository for TRU waste. The specie of NO3- is an oxidizing agent which can be reduced to NO2- and NH3/NH4+ by the coexistence of reducing materials such as metals.In order to estimate the safety of the repository for TRU waste, the impacts of nitrate on the disposal system for TRU waste have to be estimated. Especially, NH3 can elevate the solubility and reduce Rd value through the formation of ammine complexes. The quantitative information of chemical evolution of nitrate is necessary for evaluating the impact of nitrate and the chemical species arising from nitrate on the safety of the repository of TRU waste. The evolution of chemical form of NO3- by the reducing reaction accompanied with metal corrosion was experimentally examined. The reduction of NO3- was considered to be a serial reaction, that is, NO3- - NO2- - NH3. The rate equations of cathodic reactions (water reduction and reduction of nitrate and nitrite) were experimentally determined through the electrochemical measurements. The rate equation of metal dissolution (anodic reaction) which must be balanced to the cathodic reactions in charge transfer is determined from the results of immersion tests without nitrate. The combination of the rate equations forms an assessment model of chemical evolution of NO3-. The model provided the interpretation of the results of immersion tests with nitrate.
Taniguchi, Naoki; Tateishi, Tsuyoshi*; Fukudome, Kazuyuki*; Nishimura, Tsutomu*; Kawakami, Susumu
JNC TN8400 2005-014, 36 Pages, 2005/07
It has been reported that the corrosion of carbon steel is accelerated by a contact with magnetite, which is a representative corrosion product in low oxygen environment. It is important to clear the corrosion mechanism in the presence of magnetite for long term prediction of overpack corrosion. There are two possible cathodic reactions in the presence of magnetite coupled with anodic reaction. One is reduction of Fe(III) in magnetite, and the other is hydrogen evolution reaction. If the former dominate the cathodic reaction, corrosion acceleration will be stopped with the consumption of Fe(III). While, if the latter is the main cathodic reaction, corrosion acceleration is possible to be continued for a long time. In this study, corrosion rate and hydrogen evolution behavior were investigated by the immersion test of carbon steel in contact with dummy corrosion product to contribute to understanding the corrosion mechanism.
Taniguchi, Naoki; Tateishi, Tsuyoshi*; Nishimura, Tsutomu*; Kawakami, Susumu
JNC TN8400 2003-049, 23 Pages, 2004/03
The corrosion rate and hydrogen evolution behavior of carbon steel were investigated by immersion test of carbon steel in contact with dummy corrosion product. According to this result, the severe corrosion acceleration occurs when the corrosion product contains excessive Fe(III), and the main cathodic reaction is Fe(III) reduction.
Wada, Ryutaro*; Nishimura, Tsutomu*; Nakanishi, Tomoaki*; Nakayama, Takenori*; Sakashita, Shinji*; Fujiwara, Kazuo*; Tateishi, Tsuyoshi*
JNC TJ8400 2005-001, 224 Pages, 2004/02
For repository container material of high-level radioactive waste, titanium and nickel-base alloys have been investigated as high corrosion resistance metal. In this study, the effects of environmental and material factors on hydrogen absorption of titanium were investigated experimentally. As for nickel-base allys, previous studies on corrosion behavior were serched.
Wada, Ryutaro*; Nishimura, Tsutomu*; Masuda, Kaoru*; Fujiwara, Kazuo*; Imakita, Tsuyoshi*; Tateishi, Tsuyoshi*
JNC TJ8400 2004-017, 71 Pages, 2004/02
Research on changes of nitrate by interactions with metals under the wastes disposal environment containing TRU nuclide
Wada, Ryutaro*; Nishimura, Tsutomu*; Masuda, Kaoru*; Fujiwara, Kazuo*; Imakita, Tsuyoshi*; Tateishi, Tsuyoshi*
JNC TJ8400 2004-016, 194 Pages, 2004/02
Research on Changes of Nitrate by Chemical Interactions with Metals under the wastes Disposal Environment Contaioning TRU Nucride
Wada, Ryutaro*; Nishimura, Tsutomu*; Nakanishi, Tomoaki*; Fujiwara, Kazuo*; Inoue, Takao*; Tateishi, Tsuyoshi*; Masugata, Tsuyoshi*
JNC TJ8400 2003-092, 246 Pages, 2003/02
Titanium is being studied for the high-level radioactive waste package material. Titanium has good corrosion resistance, however there is the possibility of hydrogen embrittlement with absorption of hydrogen in reducing condition. Experimental studies were performed to evaluate the hydrogen absorption behaviors of titanium in reducing condition. The failure model of the titanium overpack was also examined from the viewpoint of fracture mechanism in order to evaluate the fracture behavior of the titanium overpack caused by the hydrogen absorption. (1) Scratch test was conducted in reduce condition. The surface films on the titanium specimen were analyzed to examine the changes of the existing films and the growth phenomena of the regenerated films on the titanium specimen. (2)The long-term reaction test of the titanium specimen using the glass-seal ampoules maintaining reducing condition was conducted and analyses of the hydrogen gas generation and absorption quantitative as well as the generated film evaluation were performed. (3) Under reducing condition, the electrochemical acceleration tests of the titanium specimen were conducted. The effect of acceleration rate on the hydrogen absorption and surface film was evaluated, and the prediction as to the hydrogen absorption behavior at a natural state was also made. (4) The prediction of the maximum residual stress and the evaluation of crack growth of the titanium overpack based on the previous studies were performed. Feasibility of the modeling of fracture phenomenon with existing analysis technique was examined and the items to be developed were also discussed.
Wada, Ryutaro*; Nishimura, Tsutomu*; Masuda, Kaoru*; Fujiwara, Kazuo*; Imakita, Tsuyoshi*; Tateishi, Tsuyoshi*
JNC TJ8400 2003-080, 153 Pages, 2003/02
There exists the waste including a nitrate ion as a salt in the TRU waste materials. This nitrate ion can be transferred to the nitrite ion and/or ammonia by reducing materials such as metals in the waste disposal environment, and has the possibility to affect on the disposal environment and nuclide transfer parameters.Therefore, electrochemical tests were conducted to evaluate the reaction rate parameters of the nitrate ion and metals under the low oxygen environment. The long-term reaction test using the glass-seal vessel was also conducted to grasp precisely the nitrate ion transition reaction rate and the gas generation rate caused by the reaction of metal and the nitrate ion coexist solution. (1) Reaction rate constants under various environments were obtained performing the potentiostatic holding tests with the parameters of the solution pH, temperature, and the nitrate and nitrite ion concentrations. The formula of the nitrate ion transition reaction rate was also examined based on these obtained data. (2) Conducting the immersion tests under the environment of the low oxygen and high-pH rainfall underground water site, the long-term reaction rate data were obtained on the reaction products (ammonia, hydrogen gas etc.) of metals (carbon steel, stainless steel and zircaloy etc.) with nitrate ion. The tests under the same conditions as in the past were also conducted to evaluate the test accuracy and error range of the long-term reaction test with the glass-seal vessels.
Wada, Ryutaro*; Nishimura, Tsutomu*; Masuda, Kaoru*; Fujiwara, Kazuo*; Imakita, Tsuyoshi*; Tateishi, Tsuyoshi*
JNC TJ8400 2003-079, 252 Pages, 2003/02
There exists the waste including a nitrate ion as a salt in the TRU waste materials. This nitrate ion can be transferred to the nitrite ion and/or ammonia by reducing materials such as metals in the waste disposal environment, and has the possibility to affect on the disposal environment and nuclide transfer parameters. Therefore, electrochemical tests were conducted to evaluate the reaction rate parameters of the nitrate ion and metals under the low oxygen environment. The long-term reaction test using the glass-seal vessel was also conducted to grasp precisely the nitrate ion transition reaction rate and the gas generation rate caused by the reaction of metal and the nitrate ion coexist solution. (1)}Reaction rate constants under various environments were obtained performing the potentiostatic holding tests with the parameters of the solution pH, temperature, and the nitrate and nitrite ion concentrations. The formula of the nitrate ion transition reaction rate was also examined based on these obtained data. (2) Conducting the immersion tests under the environment of the low oxygen and high-pH rainfall underground water site, the long-term reaction rate data were obtained on the reaction products (ammonia, hydrogen gas etc.) of metals (carbon steel, stainless steel and zircaloy etc.) with nitrate ion. The tests under the same conditions as in the past were also conducted to evaluate the test accuracy and error range of the long-term reaction test with the glass-seal vessels.
Wada, Ryutaro*; Yamaguchi, Kenji*; Nishimura, Tsutomu*; Fujiwara, Kazuo*; Tateishi, Tsuyoshi*
JNC TJ8400 2003-007, 86 Pages, 2003/02
The overpack for the high-level radioactive waste disposal is expected to maintain the long-term integrity and the corrosion behavior model of the overpack has been developed so far. To verify the adequacy and conservatism of the model, in this study, measurements of hydrogen absorption regarding hydrogen embrittlement of carbon steel, one of the candidate materials, were carried out in the compacted bentonite coexistence environment. The definite research programs to be performed in the future were also established for the important subjects as the extralong tests: (1)The hydrogen penetration-measuring device was designed to measure the penetrated hydrogen caused by the corrosion of carbon steel in the buffer material (compacted bentonite). (2)Out of generated hydrogen by the corrosion of carbon steel in the buffer material, the penetrated hydrogen volumes into carbon steel were measured. (3) Research programs on the four laboratory test themes and the three site test themes were established as the extralong test themes to be performed in the future.
Wada, Ryutaro*; Nishimura, Tsutomu*; Masuda, Kaoru*; Fujiwara, Kazuo*; Imakita, Tsuyoshi*; Tateishi, Tsuyoshi*
JNC TJ8400 2003-077, 156 Pages, 2002/02
Some TRU wastes contain nitrate ions as salt.The nitrate ions might transform into NO2- and NH3, etc. in the disposal site environment because of reducing agent such as metals, possibly changing disposal site environment or affecting nuclide migration parameters.Therefore, we investigated of chemical interaction between NO3- and metals in a low oxygen environment that corresponds to the disposal site environment.
Wada, Ryutaro*; Nishimura, Tsutomu*; Masuda, Kaoru*; Fujiwara, Kazuo*; Imakita, Tsuyoshi*; Tateishi, Tsuyoshi*
JNC TJ8400 2003-076, 300 Pages, 2002/02
Some TRU wastes contain nitrate ions as salt.The nitrate ions might transform into NO2- and NH3, etc. in the disposal site environment because of reducing agent such as metals, possibly changing disposal site environment or affecting nuclide migration parameters. Therefore, we investigated of chemical interaction between NO3- and metals in a low oxygen environment that corresponds to the disposal site environment.
Wada, Ryutaro*; Kurimoto, Yoshitaka*; Fujiwara, Kazuo*; ; Tateishi, Tsuyoshi*; Masugata, Tsuyoshi*
JNC TJ8400 2002-002, 111 Pages, 2002/02
In geologic disposal system of high-level radioactive waste, confinement by waste container must be assured over a thousand years. Titanium is one of the candidate materials, so it is important to clarify hydrogen embrittlement property under geological environment for the container lifetime prediction. The purpose of this study is to investigate hydrogen embrittlement behavior of titanium under reducing condition. Hydrogen was absorbed into titanium test pieces by electrochemical method, and tensile bending and impact tests were performed for mechanical property research. Under 1000ppm concentration of hydrogen, while distinct degradation of mechanical properties by hydrogen embrittlement occurred on dynamic stress, micro cracks induced by hydride were observed in fracture, but distinct degradation of mechanical properties by hydrogen embrittlement did not occur on static stress. Under low oxygen circumstances, corrosion rates of titanium were estimated 10 micrometer/year by hydrogen absorption method, on the contrary to 10
micrometer/year by hydrogen absorption method, on the contrary to 10 micrometer/year by gas evolution method. These results indicated hydrogen generated by corrosion of titanium under reducing condition, is almost absorbed into material. Carbon steel is regarded as reinforcement of the titanium nuclear fuel waste container. Magnetite, corrosion product of carbon steel, is considered to accelerate corrosion rate. Contribution of hydrogen evolution reaction to its acceleration is estimated to ca.60%.
micrometer/year by gas evolution method. These results indicated hydrogen generated by corrosion of titanium under reducing condition, is almost absorbed into material. Carbon steel is regarded as reinforcement of the titanium nuclear fuel waste container. Magnetite, corrosion product of carbon steel, is considered to accelerate corrosion rate. Contribution of hydrogen evolution reaction to its acceleration is estimated to ca.60%.
Wada, Ryutaro*; Yamaguchi, Kenji*; Nishimura, Tsutomu*; Kurimoto, Yoshitaka*; Nakanishi, Tomoaki*; Takeuchi, Yasunori*; Fujiwara, Kazuo*; Tateishi, Tsuyoshi*
JNC TJ8400 2002-001, 121 Pages, 2002/02
The overpack is expected to have long-term soundness, and corrosion behavior models of the overpack have been constructed. To verify validity and maintainability of the models, this study conducted a research on a super-long-term test of overpack materials for several decades. (1)We designed a testing device that would allow long-term monitoring of the corrosion behavior of carbon steel, an overpack candidate material, and the condition in the buffer material in an environment corresponding to the environment immediately after disposal. (2)We examined and prototyped a test container that would allow long-term immersion tests of carbon steel without maintenance operations such as solution exchange. (3)We proposed super-long-term tests (about 20 years) that would contribute to future geological disposal projects and safety rules. (4)We investigated a method for managing and preserving the records of the super-long-term test for the test period (20 years).
Wada, Ryutaro*; Nishimura, Tsutomu*; ; Fujiwara, Kazuo*; Tateishi, Tsuyoshi*
JNC TJ8400 2000-040, 171 Pages, 2000/02
In order to evaluate the hydrogen gas generation rate due to the corrosion of metallic materials contained in TRU wastes, the ampoule type immersion tests (the test specimen and test solution are enclosed into a glass ampoule) have been performed following the last year. The gas generation tests on zircaloy, stainless steel and carbon steel were performed under the aerobic condition in this year. (1)The hydrogen gas generation rates due to the corrosion of stainless steel (SUS304) in simulated underground water (pH8 13.5) were 0.01
13.5) were 0.01 0.15
0.15 m/y (oxygen concentration = 20%) and
m/y (oxygen concentration = 20%) and  0.1
0.1 m/y (oxygen concentration = 1%). (2)The hydrogen gas generation rate due to the corrosion of carbon steel (SPHC) in simulated underground water (pH8
m/y (oxygen concentration = 1%). (2)The hydrogen gas generation rate due to the corrosion of carbon steel (SPHC) in simulated underground water (pH8 13.5) ranged in equivalent corrosion rate of about 0.02
13.5) ranged in equivalent corrosion rate of about 0.02 12
12 m/y (oxygen concentration = 20%) and
m/y (oxygen concentration = 20%) and  0.3
0.3 m/y(oxygen concentration = 1%) depending on the composition of test solution, and the ranking by pH of solution was pH8
m/y(oxygen concentration = 1%) depending on the composition of test solution, and the ranking by pH of solution was pH8 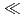 pH10
pH10  pH13.5
pH13.5  pH12.5. (3)The hydrogen gas generation rate due to the corrosion of zircaloy in simulated underground water (pH10
pH12.5. (3)The hydrogen gas generation rate due to the corrosion of zircaloy in simulated underground water (pH10 13.5) decreased with the immersion time, and the equivalent corrosion rate after immersion of 30
13.5) decreased with the immersion time, and the equivalent corrosion rate after immersion of 30 180 days was
180 days was  0.004
0.004 m/y. (4)The corrosion rates of zircaloy, stainless steel and carbon steel based on oxygen were higer than that on the hydrogen gas generation.
m/y. (4)The corrosion rates of zircaloy, stainless steel and carbon steel based on oxygen were higer than that on the hydrogen gas generation.