Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Saga, Kaname
JAEA-Review 2025-003, 23 Pages, 2025/05
Diagnosis and treatment using radioisotopes (RI) in the medical field contribute to improving people's welfare. However, almost all medical RI distributed in Japan are imported from overseas. As a result, geopolitical influences and natural disasters lead to difficulties for importing them. Based on these backgrounds, in Japan, a specialized subcommittee on the production and utilization of medical radioisotopes was established within the Atomic Energy Commission, and in May 2022, it formulated the "Action Plan for Promotion of Production and Utilization of Medical Radioisotopes." Japan Atomic Energy Agency (JAEA) launched the NXR Development Center in FY2024 to separate and recycle valuable elements contained in high-level liquid waste (HLLW). The advantages of using HLLW are that it contains a wide variety of nuclides and in large quantities. Therefore, this report focused on the RI contained in HLLW and evaluated whether it can be supplied for medical use. Specifically, the target supply amount of Sr-90, the parent nuclide of Y-90 approved as a RI for medical use, and the amount of Sr-90 in HLLW were estimated. Based on the estimation, the feasibility of separating medical RI from HLLW in a reprocessing research facility was evaluated. As a result, the HLLW possibly contains an amount of RI equivalent to the domestic medical demand. Although it depends on the RI concentration in the HLLW, a small volume of HLLW, ranging from a few hundred milliliters to a few liters, could potentially produce an amount of medical RI equivalent to domestic demand. In addition, the equipment already installed in research facilities, such as NUCEF at JAEA, may be sufficient to produce the medical RI. It may be possible to meet domestic medical demand for Sr-90, as a source of Y-90, by processing a few hundred milliliters to a few liters of HLLW using an existing research facility.
Ito, Kengo*; Takahashi, Shin*; Kato, Chizu*; Fukutani, Satoshi*; Matsumura, Tatsuro; Fujii, Toshiyuki*
Journal of Radioanalytical and Nuclear Chemistry, 334, p.2467 - 2475, 2025/02
Times Cited Count:0 Percentile:0.00(Chemistry, Analytical)In this study, the solvent extraction behavior of tin (Sn), specifically 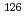 Sn, from high-level radioactive waste was evaluated using six different extractants in a HNO
Sn, from high-level radioactive waste was evaluated using six different extractants in a HNO system. Among the tested extractants, N,N-Didodecyl-2-hydroxyacetoamide (HAA) exhibited higher efficiency, still not sufficient for industrial implementation. In systems where HCl was added to HNO
system. Among the tested extractants, N,N-Didodecyl-2-hydroxyacetoamide (HAA) exhibited higher efficiency, still not sufficient for industrial implementation. In systems where HCl was added to HNO , both tributyl phosphate (TBP) and N,N,N,N'-tetra-2-ethylhexyl diglycolamide (TEHDGA) achieved D
, both tributyl phosphate (TBP) and N,N,N,N'-tetra-2-ethylhexyl diglycolamide (TEHDGA) achieved D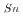 values greater than 1 at
values greater than 1 at  1 M HCl. However, due to practical challenges in industrial applications, HAA extraction in HNO
1 M HCl. However, due to practical challenges in industrial applications, HAA extraction in HNO systems, particularly at low Sn concentrations (0.0008 M), may provide a more effective solution for Sn recovery.
systems, particularly at low Sn concentrations (0.0008 M), may provide a more effective solution for Sn recovery.
Sasaki, Yuji; Kaneko, Masashi; Ban, Yasutoshi; Suzuki, Tatsuya*
Solvent Extraction Research and Development, Japan, 32(1), p.21 - 29, 2025/00
The magnitude of the masking effect of carboxylic, amic-acidic, and amidic compounds through Ln and Am extractions by tetraoctyl diglycolamide (TODGA) is compared. The compounds used are diglycol, ethylenediamine, diethylenetriamine-type, and two other amides (dioxaoctane diamide and nitrylotriacetamide). The results show that below pH 1.2, where carboxylic acids are less dissociated, amide O atoms have higher reactivity with lanthanides than O atoms in carboxyl groups. From observing the Ln patterns (D(Ln) vs. their atomic number), the compounds primarily show high reactivity, with middle and heavy Ln having a higher charge density than light Ln. Four amide compounds are employed in this work. Those with tertiary amine N atoms have pH dependence on D(Ln) due to protonation and dissociation from amine N atoms.
Tokumitsu, Shun*; Mishima, Takumi*; Matsumiya, Masahiko*; Sasaki, Yuji
Journal of Molecular Liquids, 414, Part A, p.126150_1 - 126150_8, 2024/11
The coordination states of Ln(III), (Ln=Pr, Nd, Tb and Dy) in ILs were investigated by Raman spectroscopy. The thermodynamic properties for the isomerism of [TFSA]- from trans- to cis-isomer were evaluated. The cis-[TFSA]- conformer bound to Ln3+ cation was the preferred coordination state of [Ln(III)(cis-TFSA)5]2-. The bonding energies of [Ln(III)(cis-TFSA)5]2-, (Ln=Pr, Nd, Tb and Dy) were estimated from DFT calculation.
Saga, Kaname
JAEA-Review 2024-038, 9 Pages, 2024/09
The purpose of this report is understanding the elements and radio isotopes with highly useful based on the current trends in the industrial field. The survey was conducted from the viewpoint of the abundance of elements and radio isotopes contained and the demand in the industrial field, and the following survey results were obtained. The economic scale of radio isotopes in the industrial field (including radiation use) has been increasing in recent years in the manufacturing, medical, and agricultural sectors. On the other hand, the domestic production of the utilized radio isotope is still small, and some radio isotopes are entirely imported. Radio isotopes such as Sr-90, Mo-100, Cs-137, and Am-241 from spent fuel are suitable for industrial use because of their abundance in spent fuel and half-lives. As for the utilization of elements, the industrial use of platinum group elements and rare earth elements were explored because these elements are high industrial value and low domestic self-sufficiency. The platinum group elements were evaluated to have the potential to be supplied in a certain amount as a new domestic production source based on their abundance in spent fuel. On the other hand, for rare earth elements, which have also low self-sufficiency rate, the ratio of the amount that could be supplied from spent fuel compared to the current annual supply was evaluated to be less than 1%, and therefore, no effect could be expected. The domestic recycling rate of rare earth elements is low, and the provision of numerical simulation technology, which improves the recycling rate, could highly contribute to the industries. This technology makes it possible to calculate the optimal operating conditions for the separation process, such as the number of processing stages and processing speed, in accordance with the elements to be separated and used.
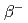 decay properties of
decay properties of  = 126, 125 nuclei; Role of Gamow-Teller and first-forbidden transitions in the half-lives
= 126, 125 nuclei; Role of Gamow-Teller and first-forbidden transitions in the half-livesKumar, A.*; Shimizu, Noritaka*; Utsuno, Yutaka; Yuan, C.*; Srivastava, P. C.*
Physical Review C, 109(6), p.064319_1 - 064319_18, 2024/06
Times Cited Count:5 Percentile:93.50(Physics, Nuclear)no abstracts in English
Osawa, Naoki*; Kim, S.-Y.*; Kubota, Masahiko*; Wu, H.*; Watanabe, So; Ito, Tatsuya; Nagaishi, Ryuji
Nuclear Engineering and Technology, 56(3), p.812 - 818, 2024/03
Times Cited Count:2 Percentile:75.80(Nuclear Science & Technology)Yamamoto, Yusuke*; Watanabe, Takahiro; Niwa, Masakazu; Shimada, Koji
JAEA-Testing 2023-003, 67 Pages, 2024/02
A long-term geosphere stability for geological disposal is evaluated by the past geological environmental changes and modern conditions. Stable hydrogen and oxygen isotope ratios ( D,
D, 
 O) of geological samples are useful information to estimate the past environmental changes and modern conditions. Recently, the thermal conversion elemental analyzer and isotope ratio mass spectrometer (TC-EA/IRMS) were installed in the Tono Geoscience Center for
O) of geological samples are useful information to estimate the past environmental changes and modern conditions. Recently, the thermal conversion elemental analyzer and isotope ratio mass spectrometer (TC-EA/IRMS) were installed in the Tono Geoscience Center for  D and
D and 
 O measurements of geological samples. In this study, we reported analytical methods of
O measurements of geological samples. In this study, we reported analytical methods of  D and
D and 
 O using international standard reference materials. In addition, evaluation tests of uncertainty by repeated analyses of the standards were performed using the TC-EA/IRMS. Furthermore, the
O using international standard reference materials. In addition, evaluation tests of uncertainty by repeated analyses of the standards were performed using the TC-EA/IRMS. Furthermore, the  D and
D and 
 O analyses by the TC- EA/IRMS were also applied to fault rock samples.
O analyses by the TC- EA/IRMS were also applied to fault rock samples.
Nishio, Katsuhisa
Isotope News, (785), p.36 - 40, 2023/02
no abstracts in English
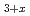 (M = Gd, Np) by the reaction of MN with Pd and chlorination of MPd
(M = Gd, Np) by the reaction of MN with Pd and chlorination of MPd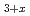 using cadmium chloride
using cadmium chlorideHayashi, Hirokazu; Shibata, Hiroki; Sato, Takumi; Otobe, Haruyoshi
Journal of Radioanalytical and Nuclear Chemistry, 332(2), p.503 - 510, 2023/02
Times Cited Count:0 Percentile:0.00(Chemistry, Analytical)The formation of MPd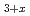 (M = Gd, Np) by the reaction of MN with Pd at 1323 K in Ar gas flow was observed. Cubic AuCu
(M = Gd, Np) by the reaction of MN with Pd at 1323 K in Ar gas flow was observed. Cubic AuCu -type GdPd
-type GdPd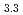 (
(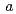 = 0.4081
= 0.4081  0.0001 nm) and NpPd
0.0001 nm) and NpPd (
(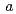 = 0.4081
= 0.4081  0.0001 nm) were identified, respectively. The product obtained from the reaction of NpN with Pd contained additional phases including the hexagonal TiNi
0.0001 nm) were identified, respectively. The product obtained from the reaction of NpN with Pd contained additional phases including the hexagonal TiNi -type NpPd
-type NpPd . Chlorination of the MPd
. Chlorination of the MPd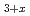 (M = Gd, Np) samples was accomplished by the solid-state reaction using cadmium chloride at 673 K in a dynamic vacuum. Pd-rich solid solution phase saturated with Cd and an intermetallic compound PdCd were obtained as by-products of MCl
(M = Gd, Np) samples was accomplished by the solid-state reaction using cadmium chloride at 673 K in a dynamic vacuum. Pd-rich solid solution phase saturated with Cd and an intermetallic compound PdCd were obtained as by-products of MCl formation.
formation.
Sato, Tetsuya; Nagame, Yuichiro*
Nihon Butsuri Gakkai-Shi, 78(2), p.64 - 72, 2023/02
The study of the chemistry of superheavy elements, which are located in the heavy extremes of the periodic table, has made considerable progress over the past 20 years, and new approaches based on various ideas have recently been developed. Research groups in Japan have also made significant contributions to the development of research on superheavy elements. Recently, notable results have been reported for the transactinide elements rutherfordium (element 104), dubnium (element 105), and seaborgium (element 106), and the heavy actinides with atomic numbers exceeding 100. The review will focus on the recent main results of these elements. This review outlines the main recent results and touches on future prospects.
Hirata, Sakiko*; Kusaka, Ryoji; Meiji, Shogo*; Tamekuni, Seita*; Okudera, Kosuke*; Hamada, Shoken*; Sakamoto, Chihiro*; Honda, Takumi*; Matsushita, Kosuke*; Muramatsu, Satoru*; et al.
Inorganic Chemistry, 62(1), p.474 - 486, 2023/01
Times Cited Count:3 Percentile:27.89(Chemistry, Inorganic & Nuclear)Uchino, Seiko*; Narita, Hirokazu*; Kita, Keisuke*; Suzuki, Hideya*; Matsumura, Tatsuro; Naganawa, Hirochika*; Sakaguchi, Koichi*; Oto, Keisuke*
Solvent Extraction Research and Development, Japan, 30(1), p.39 - 46, 2023/00
The extraction of trivalent rare earth ions (RE ) from HNO
) from HNO solution using a triamide amine, tris(N,N-di-2-ethylhexyl-ethylamide)amine (DEHTAA), was conducted, and the extraction mechanism was estimated from extraction behavior of HNO
solution using a triamide amine, tris(N,N-di-2-ethylhexyl-ethylamide)amine (DEHTAA), was conducted, and the extraction mechanism was estimated from extraction behavior of HNO and RE
and RE and the relationship between atomic number and extraction percentages (E%) for RE
and the relationship between atomic number and extraction percentages (E%) for RE . A DEHTAA molecule dominantly formed a DEHTAA HNO
. A DEHTAA molecule dominantly formed a DEHTAA HNO at 1.0 M HNO
at 1.0 M HNO and a DEHTAA(HNO
and a DEHTAA(HNO )
) at 6.0 M HNO
at 6.0 M HNO in the acid-equilibrated organic phase. This would provide the unique dependence of E% for the light RE
in the acid-equilibrated organic phase. This would provide the unique dependence of E% for the light RE on the HNO
on the HNO concentration, in which the E% value had a minimum and maximum at
concentration, in which the E% value had a minimum and maximum at  0.5 M and
0.5 M and  2 M HNO
2 M HNO , respectively. The results of the slope analyses for the distribution ratios for RE
, respectively. The results of the slope analyses for the distribution ratios for RE suggested that the dominant RE
suggested that the dominant RE complex was RE(NO
complex was RE(NO )
) DEHTAA(DEHTAA HNO
DEHTAA(DEHTAA HNO ) at 1.0 M HNO
) at 1.0 M HNO . The E% for RE
. The E% for RE decreased from La
decreased from La to Lu
to Lu at 1.0 M HNO
at 1.0 M HNO ; on the other hand, those increased from La
; on the other hand, those increased from La to Nd
to Nd at 0.25 M and from La
at 0.25 M and from La to Sm
to Sm and 6.0 M HNO
and 6.0 M HNO .
.
G tz, M.*; Yakushev, A.*; G
tz, M.*; Yakushev, A.*; G tz, S.*; Di Nitto, A.*; D
tz, S.*; Di Nitto, A.*; D llmann, Ch. E.*; Asai, Masato; Kindler, B.*; Krier, J.*; Lommel, B.*; Nagame, Yuichiro*; et al.
llmann, Ch. E.*; Asai, Masato; Kindler, B.*; Krier, J.*; Lommel, B.*; Nagame, Yuichiro*; et al.
Radiochimica Acta, 110(2), p.75 - 86, 2022/02
Times Cited Count:2 Percentile:20.20(Chemistry, Inorganic & Nuclear)The study of volatile superheavy element carbonyl complexes requires more efficient methods because the yield of transactinide elements decreases with increasing atomic number. This is achieved by using a newly developed double chamber system to separate the recoil chamber and the reaction one, thereby avoiding the decomposition of reactive molecules by the projectile ion beam, which hinders the synthesis of carbonyl complexes. The feasibility of this method was verified by synthesizing 5d metal short-lived isotopes as homologous element isotopes of the light transactinide elements Sg, Bh, Hs, and Mt at the Japan Atomic Energy Agency tandem accelerator and conducting model experiments.
Ito, Tatsuya; Osugi, Haruka*; Osawa, Naoki*; Takahashi, Tadayuki*; Kim, S.-Y.*; Nagaishi, Ryuji
Analytical Sciences, 38(1), p.91 - 97, 2022/01
Times Cited Count:4 Percentile:21.44(Chemistry, Analytical)A novel ionic liquid (IL) functionalized with thiodiglycol amic acid containing a soft S donor was synthesized for the effective and efficient extraction of platinum group metals (Ru, Rh, and Pd) from aqueous nitric acid solutions, such as high-level radioactive liquid waste (HLLW). The IL allowed a rapid extraction of Pd(II) with an extraction ratio of approximately 100%. The extractions of Ru(III) and Rh(III) by the IL were slower than that of Pd(II), but the rates were accelerated by temperature elevation. The extractions of Ru(III) and Rh(III) at 50 C reached equilibrium within 4 and 8 h, respectively, with the extraction ratios of over 90% without assisting agents or other methods for the extraction system. Furthermore, the IL could extract more than 90% of Ru(III), Rh(III), and Pd(II) from the simulated HLLW within 2 h at 50
C reached equilibrium within 4 and 8 h, respectively, with the extraction ratios of over 90% without assisting agents or other methods for the extraction system. Furthermore, the IL could extract more than 90% of Ru(III), Rh(III), and Pd(II) from the simulated HLLW within 2 h at 50 C.
C.
Kaneko, Masashi
Nihon Genshiryoku Gakkai-Shi ATOMO , 64(1), p.30 - 34, 2022/01
, 64(1), p.30 - 34, 2022/01
Partitioning of minor actinides from rare earths is one of the most important techniques to develop group separation of high-level radioactive liquid waste. In this issue, the results of prediction of separation performance between minor actinides and rare earths observed in solvent extraction and the separation mechanism by means of using density functional theory are explained.
Sasaki, Yuji; Kaneko, Masashi; Ban, Yasutoshi; Matsumiya, Masahiko*; Nakase, Masahiko*; Takeshita, Kenji*
Separation Science and Technology, 57(16), p.2543 - 2553, 2022/00
Times Cited Count:5 Percentile:28.02(Chemistry, Multidisciplinary)The mutual separation of actinides (An) from lanthanides (Ln) using the masking agent of DTPA (diethylenetriamine-pentaacetic acid) or DTBA (diethylenetriamine-triacetic acid-bis(diethylacetamide)) in the aqueous phase through DGA extraction, referring TALSPEAK method, is focused. We investigate to obtain the same separation performance using commercially available DTPA on that using DTBA. In this work, we select lactic acid (LA) of pH buffer from 10 organic acids and ethylenediamine (ED) for the pH adjustment. Almost the same D and SF values are obtained among the conditions: TODGA-DTPA-LA-NaOH, TODGA-DTPA-LA-ED, and TODGA-DTBA-LA. The experimental results using batchwise multi-stage extractions show the average yields of Ln (La to Gd) and Am to be 3.73 and 98.1% in the aqueous phase using DGA-DTPA-LA-ED, to be 3.1 and 97.0% using DGA-DTPA-LA-NaOH, and to be 1.61 and 98.7% using DGA-DTBA-LA.
Sasaki, Yuji; Kaneko, Masashi; Matsumiya, Masahiko*; Nakase, Masahiko*; Takeshita, Kenji*
Solvent Extraction and Ion Exchange, 40(6), p.620 - 640, 2022/00
Times Cited Count:2 Percentile:12.88(Chemistry, Multidisciplinary)Owing to the chemical behavior of trivalent lanthanide and actinide ions with similar ionic radii, realizing this separation is still challenging. All lanthanides, Am, and Cm can be extracted using diglycolamide (DGA), and relatively high An/Ln separation efficiencies have been obtained using diethylenetriamine-triacetic-bisamide (DTBA). To improve the previous results as well as the separation conditions, we used organic acids for pH adjustment. The advantages of this modification included low HNO , DTBA concentrations and pH stability owing to the addition of lactic acid. Under these modified conditions, the recovery rates observed were as follows: 97.1% for Nd with the co-existence of 1.59% Am in organic phase, and 98.4% for Am with the co-existence of 2.95% Nd in aqueous phase.
, DTBA concentrations and pH stability owing to the addition of lactic acid. Under these modified conditions, the recovery rates observed were as follows: 97.1% for Nd with the co-existence of 1.59% Am in organic phase, and 98.4% for Am with the co-existence of 2.95% Nd in aqueous phase.
Nankawa, Takuya; Sekine, Yurina
Isotope News, (778), p.34 - 35, 2021/12
A high-performance adsorbent was synthesized by reaction of waste bone and sodium hydrogen carbonate. Since waste bones are inexpensive and well-supplied materials, it has the potential to be used for a wide range of decontamination and removal of harmful metals.
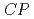 -violation studies on superheavy molecules; Theoretical and experimental perspectives
-violation studies on superheavy molecules; Theoretical and experimental perspectivesMitra, R.*; Prasannaa, V. S.*; Garcia Ruiz, R. F.*; Sato, Tetsuya; Abe, Minori*; Sakemi, Yasuhiro*; Das, B. P.*; Sahoo, B. K.*
Physical Review A, 104(6), p.062801_1 - 062801_9, 2021/12
Times Cited Count:3 Percentile:24.56(Optics)We provide detailed theoretical studies of quantities relevant to the electron electric dipole moment (eEDM) and nucleus-electron scalar-pseudoscalar interactions in diatomic molecules containing superheavy lawrencium nuclei. The sensitivity to parity and time (or, equivalently, 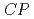 ) reversal violating properties is studied for different neutral and ionic molecules. The effective electric fields in these systems are found to be about 3
) reversal violating properties is studied for different neutral and ionic molecules. The effective electric fields in these systems are found to be about 3 4 times larger than other known molecules on which eEDM experiments are being performed. Similarly, these superheavy molecules exhibit an enhancement of more than 3 times for
4 times larger than other known molecules on which eEDM experiments are being performed. Similarly, these superheavy molecules exhibit an enhancement of more than 3 times for 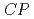 -violating scalar-pseudoscalar nucleus-electron interactions. Our preliminary analysis using the Woods-Saxon nuclear model also demonstrates that these results are sensitive to the diffuse surface interactions inside the Lr nucleus.
-violating scalar-pseudoscalar nucleus-electron interactions. Our preliminary analysis using the Woods-Saxon nuclear model also demonstrates that these results are sensitive to the diffuse surface interactions inside the Lr nucleus.