Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kobata, Masaaki; Okane, Tetsuo; Kobayashi, Keisuke*
Bunko Kenkyu, 67(4), p.161 - 162, 2018/08
We introduce hard X-ray photoelectron spectroscopy, which has been rapidly introduced and developed in synchrotron radiation facilities. In particular, in order to realize electronic state analysis by hard X-ray photoelectron spectroscopy of insulators, the developed charge neutralization method was described. As an example, we showed adsorption behavior of cesium to nuclear reactor structure assuming Fukushima Daiichi Nuclear Power Plant accident. Finally, future prospects of hard X-ray photoelectron spectroscopy will be described.
Kobata, Masaaki; Okane, Tetsuo; Nakajima, Kunihisa; Suzuki, Eriko; Owada, Kenji; Kobayashi, Keisuke*; Yamagami, Hiroshi; Osaka, Masahiko
Journal of Nuclear Materials, 498, p.387 - 394, 2018/01
Times Cited Count:17 Percentile:86.67(Materials Science, Multidisciplinary)In this study, for the understandings of Cesium (Cs) adsorption behavior on structure materials in severe accidents at a light water nuclear reactor, the chemical state of Cs and its distribution on the surface of SUS304 stainless steel (SS) with different Si concentration were investigated by hard X-ray photoelectron spectroscopy (HAXPES) and scanning electron microscope / energy dispersive X-ray spectroscopy (SEM/EDX). As a result, it was found that Cs is selectively adsorbed at the site where Si distributes with high concentration. CsFeSiO is a dominant Cs products in the case of low Si content, mainly formed, while Cs
is a dominant Cs products in the case of low Si content, mainly formed, while Cs Si
Si O
O and Cs
and Cs Si
Si O
O are formed in addition to CsFeSiO
are formed in addition to CsFeSiO in the case of high Si content. The chemical forms of the Cs compounds produced in the adsorption process on the SS surface has a close correlation with the concentration and chemical states of Si originally included in SS.
in the case of high Si content. The chemical forms of the Cs compounds produced in the adsorption process on the SS surface has a close correlation with the concentration and chemical states of Si originally included in SS.
 -Ti
-Ti O
O and
and  -Ti
-Ti O
O studied by hard X-ray photoelectron spectroscopy
studied by hard X-ray photoelectron spectroscopyKobayashi, Keisuke*; Taguchi, Munetaka*; Kobata, Masaaki; Tanaka, Kenji*; Tokoro, Hiroko*; Daimon, Hiroshi*; Okane, Tetsuo; Yamagami, Hiroshi; Ikenaga, Eiji*; Okoshi, Shinichi*
Physical Review B, 95(8), p.085133_1 - 085133_7, 2017/02
Times Cited Count:15 Percentile:56.08(Materials Science, Multidisciplinary) studied by photoelectron spectroscopy
studied by photoelectron spectroscopyKobata, Masaaki; Fujimori, Shinichi; Takeda, Yukiharu; Okane, Tetsuo; Saito, Yuji; Kobayashi, Keisuke*; Yamagami, Hiroshi; Nakamura, Ai*; Hedo, Masato*; Nakama, Takao*; et al.
Journal of the Physical Society of Japan, 85(9), p.094703_1 - 094703_6, 2016/09
Times Cited Count:13 Percentile:62.55(Physics, Multidisciplinary)Okane, Tetsuo; Kobata, Masaaki; Sato, Isamu*; Kobayashi, Keisuke*; Osaka, Masahiko; Yamagami, Hiroshi
Nuclear Engineering and Design, 297, p.251 - 256, 2016/02
Times Cited Count:2 Percentile:19.71(Nuclear Science & Technology)Kobata, Masaaki; Kobayashi, Keisuke*
Journal of the Vacuum Society of Japan, 58(2), p.43 - 49, 2015/02
We report the applications of a hard X-ray photoelectron spectroscopy to the characterization of SiO /Si(001)systems. Large escape depth of high-energy photoelectron enables us to probe buried layers and their interfaces in multilayer structures. Estimation of SiO
/Si(001)systems. Large escape depth of high-energy photoelectron enables us to probe buried layers and their interfaces in multilayer structures. Estimation of SiO overlayer thicknesses up to 25 nm by angle resolved XPS was possible in SiO
overlayer thicknesses up to 25 nm by angle resolved XPS was possible in SiO /Si(001) samples. Determination of the thickness profile of a wedged shape SiO
/Si(001) samples. Determination of the thickness profile of a wedged shape SiO buried layer was successfully done in Ir (8 nm)/HfO
buried layer was successfully done in Ir (8 nm)/HfO (2.2 nm)/thickness graded-SiO
(2.2 nm)/thickness graded-SiO (0-10 nm) / Si (100). The Si 1s core level showed a SiO
(0-10 nm) / Si (100). The Si 1s core level showed a SiO thickness dependent shift, which was ascribed to fixed charge at the SiO
thickness dependent shift, which was ascribed to fixed charge at the SiO -Si interface. Energy distribution of interface states at ultrathin thermal oxide/Si(100) interfaces were determined by Si 1
-Si interface. Energy distribution of interface states at ultrathin thermal oxide/Si(100) interfaces were determined by Si 1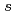 core level shift by applying gate bias in metal-oxide-semiconductor (MOS) structure with 5 nm Au gate electrodes.
core level shift by applying gate bias in metal-oxide-semiconductor (MOS) structure with 5 nm Au gate electrodes.
 nanocomposite electrocatalyst; An in situ electrochemical X-ray absorption fine structure study
nanocomposite electrocatalyst; An in situ electrochemical X-ray absorption fine structure studyMasuda, Takuya*; Fukumitsu, Hitoshi*; Fugane, Keisuke*; Togasaki, Hirotaka*; Matsumura, Daiju; Tamura, Kazuhisa; Nishihata, Yasuo; Yoshikawa, Hideki*; Kobayashi, Keisuke*; Mori, Toshiyuki*; et al.
Journal of Physical Chemistry C, 116(18), p.10098 - 10102, 2012/05
Times Cited Count:118 Percentile:93.76(Chemistry, Physical)In situ electrochemical X-ray absorption fine structure (XAFS) measurements were performed at the Pt L and Ce L
and Ce L edges of the Pt-CeO
edges of the Pt-CeO /C catalyst, which was prepared by a combined process of precipitation and coimpregnation methods, as well as at the Pt L
/C catalyst, which was prepared by a combined process of precipitation and coimpregnation methods, as well as at the Pt L edge of the conventional Pt/C catalyst in oxygen-saturated H
edge of the conventional Pt/C catalyst in oxygen-saturated H SO
SO solution to clarify the role of CeO
solution to clarify the role of CeO in the reduction of the overpotential for the oxygen reduction reaction (ORR) at the Pt-CeO
in the reduction of the overpotential for the oxygen reduction reaction (ORR) at the Pt-CeO nanocomposite compared with the conventional Pt/C catalyst. XAFS measurements clearly show that the enhancement of ORR activity is attributed to the inhibition of Pt oxide formation by the CeO
nanocomposite compared with the conventional Pt/C catalyst. XAFS measurements clearly show that the enhancement of ORR activity is attributed to the inhibition of Pt oxide formation by the CeO layer, of which Ce
layer, of which Ce was oxidized to Ce
was oxidized to Ce instead of Pt at the Pt oxide formation potential.
instead of Pt at the Pt oxide formation potential.
Kobayashi, Masaki*; Niwa, Hideharu*; Harada, Yoshihisa*; Horiba, Koji*; Oshima, Masaharu*; Ofuchi, Hironori*; Terakura, Kiyoyuki*; Ikeda, Takashi; Koshigoe, Yuka*; Ozaki, Junichi*; et al.
Journal of Power Sources, 196(20), p.8346 - 8351, 2011/10
Times Cited Count:32 Percentile:67.33(Chemistry, Physical)The electronic structure of Co atoms in CoPc-based carbon catalysts, which were prepared by pyrolyzing a mixture of CoPc and phenol resin polymer up to 1000 C, has been investigated using XAFS analysis and HXPES. The Co K XAFS spectra show that most of the Co atoms are in the metallic state and small quantities of oxidized Co components are present in the samples even after acid washing to remove Co atoms. Based on the difference in probing depth between XAFS and HXPES, it was found that after acid washing, the surface region with the aggregated Co clusters is primarily composed of metallic Co. Since the electrochemical properties remain almost unchanged even after the acid washing process, the residual metallic and oxidized Co atoms themselves will hardly contribute to the ORR activity of the CoPc-based carbon cathode catalysts, implying that the active sites of the CoPc-based catalysts primarily consist of light elements such as C and N.
C, has been investigated using XAFS analysis and HXPES. The Co K XAFS spectra show that most of the Co atoms are in the metallic state and small quantities of oxidized Co components are present in the samples even after acid washing to remove Co atoms. Based on the difference in probing depth between XAFS and HXPES, it was found that after acid washing, the surface region with the aggregated Co clusters is primarily composed of metallic Co. Since the electrochemical properties remain almost unchanged even after the acid washing process, the residual metallic and oxidized Co atoms themselves will hardly contribute to the ORR activity of the CoPc-based carbon cathode catalysts, implying that the active sites of the CoPc-based catalysts primarily consist of light elements such as C and N.
 studied by photoemission and soft X-ray absorption spectroscopies
studied by photoemission and soft X-ray absorption spectroscopiesUtsumi, Yuki*; Sato, Hitoshi*; Kurihara, Hidenao*; Maso, Hiroyuki*; Hiraoka, Koichi*; Kojima, Kenichi*; Tobimatsu, Komei*; Okochi, Takuo*; Fujimori, Shinichi; Takeda, Yukiharu; et al.
Physical Review B, 84(11), p.115143_1 - 115143_7, 2011/09
Times Cited Count:11 Percentile:44.66(Materials Science, Multidisciplinary)We have studied conduction-band (CB) electronic states of a typical valence-transition compound YbInCu by means of temperature-dependent hard X-ray photoemission spectroscopy (HX-PES), soft X-ray absorption spectroscopy (XAS), and soft X-ray photoemission spectroscopy (SX-PES) of the valence band. We have described the valence transition in YbInCu
by means of temperature-dependent hard X-ray photoemission spectroscopy (HX-PES), soft X-ray absorption spectroscopy (XAS), and soft X-ray photoemission spectroscopy (SX-PES) of the valence band. We have described the valence transition in YbInCu in terms of the charge transfer from the CB to Yb 4
in terms of the charge transfer from the CB to Yb 4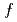 states.
states.
Niwa, Hideharu*; Kobayashi, Masaki*; Horiba, Koji*; Harada, Yoshihisa*; Oshima, Masaharu*; Terakura, Kiyoyuki*; Ikeda, Takashi; Koshigoe, Yuka*; Ozaki, Junichi*; Miyata, Seizo*; et al.
Journal of Power Sources, 196(3), p.1006 - 1011, 2011/02
Times Cited Count:90 Percentile:91.52(Chemistry, Physical)We report on the electronic structure of three different types of N-containing carbon-based cathode catalysts for polymer electrolyte fuel cells observed by hard X-ray photoemission spectroscopy. C 1s spectra show the importance of 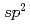 carbon network formation for the oxygen reduction reaction (ORR) activity. Samples having high oxygen reduction reaction activity in terms of oxygen reduction potential contain high concentration of graphite-like nitrogen. Based on a quantitative analysis of our results, the oxygen reduction reaction activity of the carbon-based cathode catalysts will be improved by increasing concentration of graphite-like nitrogen in a developed
carbon network formation for the oxygen reduction reaction (ORR) activity. Samples having high oxygen reduction reaction activity in terms of oxygen reduction potential contain high concentration of graphite-like nitrogen. Based on a quantitative analysis of our results, the oxygen reduction reaction activity of the carbon-based cathode catalysts will be improved by increasing concentration of graphite-like nitrogen in a developed 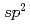 carbon network.
carbon network.
 species
speciesKobayashi, Nobuo*; Sasaki, Shinichiro*; Tasaki, Keisuke*; Nakatsuka, Akira*; Nozawa, Shigeki; Hase, Yoshihiro; Narumi, Issei
JAEA-Review 2010-065, JAEA Takasaki Annual Report 2009, P. 67, 2011/01
 Co
Co O
OKobayashi, Masaki*; Ishida, Yukiaki*; Hwang, J. I.*; Osafune, Yoshitaka*; Fujimori, Atsushi*; Takeda, Yukiharu; Okane, Tetsuo; Saito, Yuji; Kobayashi, Keisuke*; Saeki, Hiromasa*; et al.
Physical Review B, 81(7), p.075204_1 - 075204_7, 2010/02
Times Cited Count:19 Percentile:61.85(Materials Science, Multidisciplinary) using ion beam irradiation
using ion beam irradiationKobayashi, Nobuo*; Kano, Sayaka*; Sasaki, Shinichiro*; Tasaki, Keisuke*; Nakatsuka, Akira*; Nozawa, Shigeki; Hase, Yoshihiro; Narumi, Issei
JAEA-Review 2009-041, JAEA Takasaki Annual Report 2008, P. 77, 2009/12
 Mn
Mn As studied by X-ray magnetic circular dichroism
As studied by X-ray magnetic circular dichroismTakeda, Yukiharu; Kobayashi, Masaki*; Okane, Tetsuo; Okochi, Takuo*; Okamoto, Jun*; Saito, Yuji; Kobayashi, Keisuke*; Yamagami, Hiroshi; Fujimori, Atsushi*; Tanaka, Arata*; et al.
Hoshako, 22(4), p.202 - 209, 2009/07
no abstracts in English
Kobayashi, Yasuhiko; Funayama, Tomoo; Hamada, Nobuyuki*; Sakashita, Tetsuya; Konishi, Teruaki*; Imaseki, Hitoshi*; Yasuda, Keisuke*; Hatashita, Masanori*; Takagi, Keiichi*; Hatori, Satoshi*; et al.
Journal of Radiation Research, 50(Suppl.A), p.A29 - A47, 2009/03
Times Cited Count:38 Percentile:72.75(Biology)Kobayashi, Nobuo*; Tasaki, Keisuke*; Kano, Sayaka*; Sakamoto, Sakiko*; Nakatsuka, Akira*; Hase, Yoshihiro; Narumi, Issei
JAEA-Review 2008-055, JAEA Takasaki Annual Report 2007, P. 70, 2008/11
no abstracts in English
 Mn
Mn As studied by X-ray magnetic circular dichroism
As studied by X-ray magnetic circular dichroismTakeda, Yukiharu; Kobayashi, Masaki*; Okane, Tetsuo; Okochi, Takuo; Okamoto, Jun*; Saito, Yuji; Kobayashi, Keisuke*; Yamagami, Hiroshi; Fujimori, Atsushi; Tanaka, Arata*; et al.
Physical Review Letters, 100(24), p.247202_1 - 247202_4, 2008/06
Times Cited Count:39 Percentile:82.16(Physics, Multidisciplinary)no abstracts in English
 Cr
Cr Te
TeKobayashi, Masaki*; Ishida, Yukiaki*; Hwang, J. I.*; Song, G. S.*; Fujimori, Atsushi; Yang, C. S.*; Lee, L.*; Lin, H.-J.*; Huang, D.-J.*; Chen, C. T.*; et al.
New Journal of Physics (Internet), 10, p.055011_1 - 055011_15, 2008/05
Times Cited Count:15 Percentile:64.38(Physics, Multidisciplinary) Ru
Ru O
O thin films
thin filmsTerai, Kota*; Yoshii, Kenji; Takeda, Yukiharu; Fujimori, Shinichi; Saito, Yuji; Owada, Kenji; Inami, Toshiya; Okane, Tetsuo; Arita, Masashi*; Shimada, Kenya*; et al.
Physical Review B, 77(11), p.115128_1 - 115128_6, 2008/03
Times Cited Count:15 Percentile:54.78(Materials Science, Multidisciplinary) Sr
Sr TiO
TiO films by in situ resonant photoemission spectroscopy
films by in situ resonant photoemission spectroscopyLin, Y.-H.*; Terai, Kota*; Wadachi, Hiroki*; Kobayashi, Masaki*; Takizawa, Masaru*; Hwang, J. I.*; Fujimori, Atsushi; Nan, C.-W.*; Li, J.-F.*; Fujimori, Shinichi; et al.
Applied Physics Letters, 90(22), p.222909_1 - 222909_3, 2007/05
Times Cited Count:6 Percentile:26.43(Physics, Applied)Epitaxial Ba Sr
Sr TiO
TiO thin films were prepared on Nb-doped SrTiO
thin films were prepared on Nb-doped SrTiO (100) substrates by the pulsed laser deposition technique and were studied by measuring the Ti 2p
(100) substrates by the pulsed laser deposition technique and were studied by measuring the Ti 2p  3d resonant photoemission spectra in the valence-band region, both at room temperature and low temperature. The results demonstrated an abrupt variation in the spectral structures between 2.8 nm (
3d resonant photoemission spectra in the valence-band region, both at room temperature and low temperature. The results demonstrated an abrupt variation in the spectral structures between 2.8 nm ( 7 ML) and 2.0 nm (
7 ML) and 2.0 nm ( 5 ML) Ba
5 ML) Ba Sr
Sr TiO
TiO films, suggesting that there exists a critical thickness for phase change in the range of 2.0-2.8 nm.
films, suggesting that there exists a critical thickness for phase change in the range of 2.0-2.8 nm.