Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
荒井 陽一; 渡部 創; 渡部 雅之; 新井 剛*; 勝木 健太*; 吾郷 友宏*; 藤川 寿治*; 武田 啓佑*; 福元 博基*; 保科 宏行*; et al.
Nuclear Instruments and Methods in Physics Research B, 554, p.165448_1 - 165448_10, 2024/09
被引用回数:0 パーセンタイル:0.00(Instruments & Instrumentation)Radioactive aqueous and organic liquid wastes contaminated by U and radioactive materials have been generated through solvent extraction experiments on nuclear fuel materials. Although incineration and denitrification / conversion processes are promising for treatment of such liquid wastes, installation of large equipment is essential. Treatment of liquid wastes generated from the reprocessing experiments is one of important tasks of Systematic Treatments of RAdioactive liquid wastes for Decommissioning (STRAD) project, and the recovery technologies of nuclear materials from the spent solvent has been developed. However, recovery of trace amounts of nuclear fuel material from aqueous solutions with wide pH range is still a challenging task. In our previous study, the porous silica particles with a high specific surface area bearing the iminodiacetic acid (IDA) functional group were revealed to be applicable to recover cations. Although the IDA group introduced adsorbents showed an excellent adsorption reaction from the aqueous solution, further improvement related to the adsorption amount is indispensable for application to the radioactive liquid treatment. In this study, fluorous ligands with IDA group were newly synthesized, and its complexation behavior with cations was investigated in order to understand the adsorption mechanism.
廃炉環境国際共同研究センター; 信州大学*
JAEA-Review 2023-053, 87 Pages, 2024/05
日本原子力研究開発機構(JAEA)廃炉環境国際共同研究センター(CLADS)では、令和4年度英知を結集した原子力科学技術・人材育成推進事業(以下、「本事業」という)を実施している。本事業は、東京電力ホールディングス株式会社福島第一原子力発電所の廃炉等をはじめとした原子力分野の課題解決に貢献するため、国内外の英知を結集し、様々な分野の知見や経験を、従前の機関や分野の壁を越えて緊密に融合・連携させた基礎的・基盤的研究及び人材育成を推進することを目的としている。平成30年度の新規採択課題から実施主体を文部科学省からJAEAに移行することで、JAEAとアカデミアとの連携を強化し、廃炉に資する中長期的な研究開発・人材育成をより安定的かつ継続的に実施する体制を構築した。本研究は、令和2年度に採択された研究課題のうち、「革新的水質浄化剤の開発による環境問題低減化技術の開拓」の令和2年度から令和4年度分の研究成果について取りまとめたものである。本研究は、東日本大震災で発生した放射性汚染水の処理問題改善に資するため、吸着性能に優れ再利用可能なストロンチウムイオン用吸着剤の開発を目指した。その結果、ストロンチウムイオンに対して優れた吸着性能を有する、再利用可能な吸着材料の開発に成功した。現行のストロンチウム用吸着材料は、極めて高価であると共に使い捨てであるため、使用済み吸着剤の廃棄量は既に膨大となっており、それら廃棄物の保管や処分方法が大きな問題となっている。そのため、今回開発した吸着材料を実装化することで、これらの問題を大きく緩和することが期待できる。また、吸着材料は一般に粉体として得られるため、バインダーを利用してカラム通水による除染に適した粒径に造粒化する必要がある。これに対し、今回開発した材料は、最初から適した粒状物として得られるところに大きな特徴があり、煩雑な造粒化工程を省くことで、製造コストの削減も期待できる。これらの結果は当初の開発目標に合致したものであり、廃炉加速化に貢献できると考えられる。これらの研究は、研究代表者の下で各研究項目間並びに廃炉環境国際共同研究センター(CLADS)等との連携を密にして進めた。また、研究実施計画を推進するための打合せや会議等を開催した。
高畠 容子; 渡部 創; 新井 剛*; 佐藤 隆博*; 柴田 淳広
Applied Radiation and Isotopes, 196, p.110783_1 - 110783_5, 2023/06
被引用回数:2 パーセンタイル:43.92(Chemistry, Inorganic & Nuclear)An adsorbent used for the recovery of trivalent minor actinides (MA(III); Am and Cm) from high level liquid waste generated from reprocessing of spent nuclear fuel was subjected to micro-PIXE analysis to improve its elution performance. The experimental adsorbent comprised SiO particles, a polymer coating, and an TODGA. The particles to be analyzed were subjected to Nd adsorption and an elution operation, but Nd in the adsorbent was found to be uniformly distributed. This might have been caused by individual differences in the amount of impregnated TODGA. The remaining Nd species were not localized to a specific part of the adsorbent after the adsorption operation. Some Nd elements were retained in the adsorbent after elution, probably because of the poor diffusion of the mobile phase inside the adsorbent. An adsorbent having a different microstructure from the first was then evaluated, and rapid elution was observed on new adsorbent along micro-PIXE analysis
particles, a polymer coating, and an TODGA. The particles to be analyzed were subjected to Nd adsorption and an elution operation, but Nd in the adsorbent was found to be uniformly distributed. This might have been caused by individual differences in the amount of impregnated TODGA. The remaining Nd species were not localized to a specific part of the adsorbent after the adsorption operation. Some Nd elements were retained in the adsorbent after elution, probably because of the poor diffusion of the mobile phase inside the adsorbent. An adsorbent having a different microstructure from the first was then evaluated, and rapid elution was observed on new adsorbent along micro-PIXE analysis
廃炉環境国際共同研究センター; 信州大学*
JAEA-Review 2022-067, 98 Pages, 2023/03
日本原子力研究開発機構(JAEA)廃炉環境国際共同研究センター(CLADS)では、令和3年度英知を結集した原子力科学技術・人材育成推進事業(以下、「本事業」という)を実施している。本事業は、東京電力ホールディングス株式会社福島第一原子力発電所の廃炉等を始めとした原子力分野の課題解決に貢献するため、国内外の英知を結集し、様々な分野の知見や経験を、従前の機関や分野の壁を越えて緊密に融合・連携させた基礎的・基盤的研究及び人材育成を推進することを目的としている。平成30年度の新規採択課題から実施主体を文部科学省からJAEAに移行することで、JAEAとアカデミアとの連携を強化し、廃炉に資する中長期的な研究開発・人材育成をより安定的かつ継続的に実施する体制を構築した。本研究は、研究課題のうち、令和2年度に採択された「革新的水質浄化剤の開発による環境問題低減化技術の開拓」の令和3年度の研究成果について取りまとめたものである。本研究は、日英国際共同研究により再利用可能なストロンチウムイオン用吸着剤を開発し、使用済み吸着剤の発生量の削減を目指すものである。令和3年度は、初年度の知見を踏まえて材料作製法の改良を行うとともに、種々の金属塩を添加剤として加え、収率や吸着性能への影響について検討を行った。得られた材料をSEM等で観察することで構造解析を行い、得られた構造解析結果と吸着性能結果とを併せて理論解析を行った。その結果、材料によっては添加剤を加えることで、吸着性能が向上する知見が得られた。また材料の再利用化条件を検討することで、再利用可能であることを見出した。これらの研究は、研究代表者の下で各研究項目間ならびにCLADS等との連携を密にして進めた。また、研究実施計画を推進するための打合せや会議等を開催した。
鈴木 智也*; 大坪 右京*; 尾形 剛志*; 塩飽 秀啓; 小林 徹; 矢板 毅; 松岡 光昭*; 村山 憲弘*; 成田 弘一*
Separation and Purification Technology, 308, p.122943_1 - 122943_7, 2023/03
被引用回数:3 パーセンタイル:15.39(Engineering, Chemical)主にAgを含む使用済み製品からPd金属をリサイクルするために硝酸抽出が行われており、ほとんどのPd残留物はAg(I)含有溶液から分離される。しかし、このAg(I)溶液中には少量のPd(II)が残存していることが多い。そのため、硝酸溶液中のPd(II)とAg(I)を分離することは、Pdのリサイクルを効率的に進めるために必要不可欠である。本研究では、アミン(R-Amine),イミノ二酢酸(R-IDA),ピリジン(R-Py),ビスピコリルアミン(R-BPA)で機能化した4種類の窒素ドナー型吸着材を用いて硝酸溶液中のPd(II), Ag(I)を分離する研究を実施した。R-Amine,R-IDA,R-Pyは硝酸溶液からAg(I), Cu(II), Ni(II), Fe(III)を選択的に吸着したが、R-AmineはPdの吸着効率が低いことが判明した。一方、R-BPAはPd(II), Ag(I), Cu(II)を全濃度において90%以上吸着することができた。フーリエ変換赤外分光法及び広域X線吸収微細構造法(EXAFS)による吸着金属イオンの構造解析から、窒素ドナー型吸着剤の分離機構を明らかにした。R-IDA, R-Py, R-BPAへのPd(II)吸着は官能基(それぞれイミノ二酢酸,ピリジン,ビスピコリルアミン)へのPd(II)配位を介して起こり、R-Amineへの吸着はNO
 と[Pd(NO
と[Pd(NO )
) ]
] のアニオン交換を介して起こることがわかった。R-IDA,R-Py,R-BPAは、配位性吸着機構により、高いPd(II)吸着挙動を示すことが明らかとなった。塩酸(5.0M)およびチオ尿素(0.1M)溶離液は、それぞれR-IDAから83%およびR-Pyから95%のPd(II)を脱離させた。R-Pyは吸着選択性と脱着効率から最も効果的なPd(II)吸着剤だった。
のアニオン交換を介して起こることがわかった。R-IDA,R-Py,R-BPAは、配位性吸着機構により、高いPd(II)吸着挙動を示すことが明らかとなった。塩酸(5.0M)およびチオ尿素(0.1M)溶離液は、それぞれR-IDAから83%およびR-Pyから95%のPd(II)を脱離させた。R-Pyは吸着選択性と脱着効率から最も効果的なPd(II)吸着剤だった。
廃炉環境国際共同研究センター; 信州大学*
JAEA-Review 2021-051, 81 Pages, 2022/01
日本原子力研究開発機構(JAEA)廃炉環境国際共同研究センター(CLADS)では、令和2年度英知を結集した原子力科学技術・人材育成推進事業(以下、「本事業」という)を実施している。本事業は、東京電力ホールディングス株式会社福島第一原子力発電所の廃炉等をはじめとした原子力分野の課題解決に貢献するため、国内外の英知を結集し、様々な分野の知見や経験を、従前の機関や分野の壁を越えて緊密に融合・連携させた基礎的・基盤的研究及び人材育成を推進することを目的としている。平成30年度の新規採択課題から実施主体を文部科学省からJAEAに移行することで、JAEAとアカデミアとの連携を強化し、廃炉に資する中長期的な研究開発・人材育成をより安定的かつ継続的に実施する体制を構築した。本研究は、研究課題のうち、令和2年度に採択された「革新的水質浄化剤の開発による環境問題低減化技術の開拓」の令和2年度の研究成果について取りまとめたものである。本研究は、日英国際共同研究により再利用可能なストロンチウムイオン用吸着剤を開発し、使用済み吸着剤の発生量の削減を目指すものである。本研究の基本的な戦略は、信州大学チーム(信州大)で作製した吸着剤を分子科学研究所チーム(分子研)と英国チームで構造解析し、その結果を信州大で得られた吸着性能と合わせて東北大学チーム(東北大)がデータ科学的に理論解析し、吸着性能の改善に向けた作製指針を導き出す。その結果を信州大にフィードバックすることで、新たな吸着剤の作製につなげていくというものである。また、吸着剤由来の廃棄物処理法についても英国チームで検討することで、吸着剤の製造から廃棄までの一貫した研究が可能である。令和2年度の成果についてであるが、信州大は予備研究で見出された材料作製法を様々な条件で検討し、得られた材料の粒径分布や機械的強度などの物性への影響を調べた。また、塩化ストロンチウム単一溶液や人工海水に塩化ストロンチウムを溶解させた模擬汚染水中のストロンチウムイオンの吸着実験を行い、ICP装置により濃度を測定して分配係数を計算し、イオン選択性を評価した。得られた材料を分子研と英国チームに送り構造解析を依頼すると共に、吸着機能結果を東北大に送りデータ解析を依頼した。
 Cs in spent Cs adsorbent used for the decontamination of radiocesium-containing water by laser ablation inductively coupled plasma mass spectrometry
Cs in spent Cs adsorbent used for the decontamination of radiocesium-containing water by laser ablation inductively coupled plasma mass spectrometry浅井 志保*; 大畑 昌輝*; 半澤 有希子; 堀田 拓摩; 蓬田 匠; 北辻 章浩
Analytical Chemistry, 92(4), p.3276 - 3284, 2020/02
被引用回数:7 パーセンタイル:30.21(Chemistry, Analytical)福島第一原子力発電所の汚染水処理に使用されたCs吸着材を安全に処分するために、長寿命核種である Csの放射能を把握する必要がある。
Csの放射能を把握する必要がある。 Csは、誘導結合プラズマ質量分析計(ICP-MS)で測定するが、通常、液体試料のみに対応しているため、廃Cs吸着材の場合、Csの溶離操作が不可欠となる。しかし、
Csは、誘導結合プラズマ質量分析計(ICP-MS)で測定するが、通常、液体試料のみに対応しているため、廃Cs吸着材の場合、Csの溶離操作が不可欠となる。しかし、 Csから放出される強い放射線が取り扱いを困難にする。そこで本研究では、固体試料の直接測定が可能なレーザーアブレーションICP-MSを用いて
Csから放出される強い放射線が取り扱いを困難にする。そこで本研究では、固体試料の直接測定が可能なレーザーアブレーションICP-MSを用いて Cs/
Cs/ Csを測定し、
Csを測定し、 Csの
Csの 線測定結果と合わせて、Cs吸着材中の
線測定結果と合わせて、Cs吸着材中の Csを簡便かつ精確に定量する方法を開発した。方法の妥当性を確認するため、放射性セシウムを含む汚染水に市販のCs吸着材を浸漬させて模擬試料を調製し測定したところ、水試料の分析値と一致した。
Csを簡便かつ精確に定量する方法を開発した。方法の妥当性を確認するため、放射性セシウムを含む汚染水に市販のCs吸着材を浸漬させて模擬試料を調製し測定したところ、水試料の分析値と一致した。
 -P adsorbent for MA(III) recovery
-P adsorbent for MA(III) recovery小藤 博英; 渡部 創; 竹内 正行; 鈴木 英哉; 松村 達郎; 塩飽 秀啓; 矢板 毅
Progress in Nuclear Science and Technology (Internet), 5, p.61 - 65, 2018/11
Applicability of newly developed MA(III)/RE(III) separation extractant TPDN for the extractionchromatography technology was evaluated through series of fundamental characterizations. The adsorbentselectively extract  Am from diluted high level liquid waste and sufficient performance for MA(III)/RE(III)separation process was confirmed. Durability and behavior in the vitrification process of the adsorbent alsosuggested that the new adsorbent can be employed as a material for reprocessing of spent fuels.
Am from diluted high level liquid waste and sufficient performance for MA(III)/RE(III)separation process was confirmed. Durability and behavior in the vitrification process of the adsorbent alsosuggested that the new adsorbent can be employed as a material for reprocessing of spent fuels.
竹内 正行; 佐野 雄一; 渡部 創; 中原 将海; 粟飯原 はるか; 小藤 博英; 小泉 務; 水野 朋保
Proceedings of 2017 International Congress on Advances in Nuclear Power Plants (ICAPP 2017) (CD-ROM), 6 Pages, 2017/04
SmART cycle test has been promoted for reduction of volume and radiotoxicity of vitrified waste. It is a fuel cycle test using small amount of minor actinides (MA) in irradiated FBR fuels. The plan includes U, Pu and MA partitioning, fuel fabrication, irradiation at FBR and post irradiation examination. In this paper, a series of radioactive tests for MA partitioning from the irradiated fuel were mainly focused. As treatment of the irradiated fuel, it was sheared and dissolved by hot nitric acid, and then, U, Pu and Np in the dissolved fuel solution are co-extracted by solvent extraction. Am and Cm in the raffinate are efficiently separated by chromatographic technique. The target of MA yield for this cycle test is more than 1g to fabricate MOX pellets bearing 5% MA. As the current status for the MA partitioning, we have successfully finished the shearing and dissolution of the irradiated FBR fuel and solvent extraction process and the two steps flowsheet for Am and Cm partitioning from the raffinate and denitration behavior of separation product solution was discussed.
Awual, M. R.; 宮崎 有史; 田口 富嗣; 塩飽 秀啓; 矢板 毅
Chemical Engineering Journal, 291, p.128 - 137, 2016/05
被引用回数:246 パーセンタイル:99.19(Engineering, Environmental)A novel macrocyclic ligand of di-o-benzo-p-xylyl-28-crown-8-ether (DOBPX28C8) was prepared and successfully anchored onto the mesoporous silica for the fabrication of mesoporous hybrid adsorbent (MHA) using direct immobilization approach and serving as an efficient adsorbent for Cs removal. The high amount of K (7.69 mM) was slightly affected the Cs adsorption because the hydration radius of K is close to the Cs than that of the other cation of Na. Also the Cs adsorption efficiency was more higher even in the presence of 1,025 fold K and 1,739 fold Na, respectively. The functional group of DOBPX28C8 ligand embedded onto the mesoporous silica exhibited more conformational freedom, and the benzene ring in para position was consolidated for pai-electron orientation with expanding the ring size of DOBPX28C8 for easy capturing with high selectivity and adsorption ability towards the Cs.
Awual, M. R.; 矢板 毅; 宮崎 有史; 松村 大樹; 塩飽 秀啓; 田口 富嗣
Scientific Reports (Internet), 6, p.19937_1 - 19937_10, 2016/01
被引用回数:201 パーセンタイル:97.15(Multidisciplinary Sciences)A novel macrocyclic ligand of 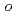 -benzo-
-benzo- -xylyl-22-crown-6-ether (OBPX22C6) was developed and successfully immobilized onto mesoporous silica for the preparation of hybrid adsorbent. The benzene ring
-xylyl-22-crown-6-ether (OBPX22C6) was developed and successfully immobilized onto mesoporous silica for the preparation of hybrid adsorbent. The benzene ring  electron is the part of crown ether of OBPX22C6 for easy orientation of the macrocyclic compound for making the
electron is the part of crown ether of OBPX22C6 for easy orientation of the macrocyclic compound for making the  electron donation with Cs complexation. The results clarified that the Cs removal process was rapid and reached saturation within a short time. Considering the effect of competitive ions, sodium did not markedly affect the Cs adsorption whereas potassium was slightly affected due to the similar ionic radii. Due to its high selectivity and reusability, significant volume reduction is expected as this promising hybrid adsorbent is used for Cs removal in Fukushima wastewater.
electron donation with Cs complexation. The results clarified that the Cs removal process was rapid and reached saturation within a short time. Considering the effect of competitive ions, sodium did not markedly affect the Cs adsorption whereas potassium was slightly affected due to the similar ionic radii. Due to its high selectivity and reusability, significant volume reduction is expected as this promising hybrid adsorbent is used for Cs removal in Fukushima wastewater.
Awual, M. R.; Eldesoky, G. E.*; 矢板 毅; Naushad, M.*; 塩飽 秀啓; Alothman, Z. A.*; 鈴木 伸一
Chemical Engineering Journal, 279, p.639 - 647, 2015/11
被引用回数:260 パーセンタイル:99.16(Engineering, Environmental)A novel Schiff base ligand based nano-composite adsorbent (NCA) was prepared for the detection and removal of copper (Cu(II)) ions in wastewater samples. Upon the addition of Cu(II) ions to NCA at optimum conditions, the clear color was visible to the naked-eye in the detection system. This NCA exhibited an obvious color change from yellowish to dark green in the presence of Cu(II) ions in aqueous solution. The NCA could detect the Cu(II) ions over other foreign ions with high sensitivity and selectivity. For adsorption behaviour, influences several factors such as solution pH, contact time, concentration for Cu(II) ion adsorption was investigated by batch experiment in detail. The results showed that neutral solution pH was suitable to get optimum Cu(II) ions adsorption.
Awual, M. R.; 矢板 毅; 塩飽 秀啓; 鈴木 伸一
Chemical Engineering Journal, 276, p.1 - 10, 2015/09
被引用回数:194 パーセンタイル:98.41(Engineering, Environmental)The organic ligand of N,N(octane-1,8-diylidene)di(2-hydroxyl-3,5-dimethylaniline) was synthesized and anchored onto inorganic mesoporous materials by direct immobilization method for Co(II) ions capturing from waste samples. The interaction of this material with various metal ions was evaluated, and it exhibited distinct color change from whitish to green in the presence of Co(II) ions, detectable even to the naked-eye. The effects of solution pH, contact time, competing ions and initial concentration on Co(II) sorption on NCA were investigated under optimum conditions. The data also clarified that the NCA adsorbent is an efficient and eco-friendly adsorbent for Co(II) treatment. Therefore, the proposed adsorbent can be considered as a potential candidate for Co(II) ions monitoring and removal from wastewater in large-scale operations.
高畠 容子; 柴田 淳広; 野村 和則
Proceedings of 21st International Conference & Exhibition; Nuclear Fuel Cycle for a Low-Carbon Future (GLOBAL 2015) (USB Flash Drive), p.2099 - 2104, 2015/09
The radioactive contaminated water has accumulated at Fukushima Dai-ichi Nuclear Power Station after the severe damage by the events accompanying the earthquake and tsunami. The concentration of Ca in the accumulated water is higher than that of Sr. Because of their chemical similarity, the secondary radioactive waste from Sr decontamination process will contains Ca more than Sr. Therefore, the volume of the secondary waste will increase more unnecessarily. Three kinds of Sr adsorbents; SW-KAZLS, the Hydrous titanic acid and SrTreat, were characterized and assessed from the viewpoint of Sr decontamination for radioactive contaminated water. The amount of Sr in SW-KAZLS showed the highest Sr selectivity. Since the Hydrous titanic acid was the hardest adsorbent of three, it is applied well to adsorption tower. The amount of Sr in SrTreat was favorable, and it could be applied to decontamination for the radioactive contaminated water of low Sr concentration.
Shahat, A.*; Awual, M. R.; Khaleque, M. A.*; Alam, M. Z.*; Naushad, M.*; Chowdhury, A. M. S*
Chemical Engineering Journal, 273, p.286 - 295, 2015/08
被引用回数:349 パーセンタイル:99.57(Engineering, Environmental)The detection and removal of lead (Pb(II)) ions in water is essential for both human health and the natural environment. This study reported the feasibility of the large-pore diameter nano-adsorbent for capturing low level Pb(II) ions from aqueous solutions in a batch system, in order to comply with the toxicity limit for discharging wastewaters. The proposed Pb(II) detection system has good characteristics with high sensitivity and selectivity, robustness, easy operation, portability and use of a small sample volume. The competing ions effect was assessed with spiked samples and the nano-adsorbent exhibited high selectivity to Pb(II) ions with optimum color formation and the detection procedure can be easily applied to rapid on-site Pb(II) ions monitoring.
Shahat, A.*; Awual, M. R.; Naushad, M.*
Chemical Engineering Journal, 271, p.155 - 163, 2015/07
被引用回数:250 パーセンタイル:99.09(Engineering, Environmental)Ligand anchored functional nanomaterials are increasingly gaining interest as efficient materials for various types of toxic pollutants. In this study, the organic ligand was densely anchored onto the mesoporous silica by the building-block approach as facial adsorbent, characterized and then examined for the cobalt (Co(II)) ion detection and removal from aqueous solutions. This study also examined the possibility of using facial adsorbent to remove low concentrations of Co(II) ions from water solution. The sorption isotherms fit the Langmuir sorption model well and the maximum Co(II) ion sorption capacity was 157.73 mg/g. The effective eluent of 0.20M HCl was used to elute the Co(II) from the facial adsorbent, and the adsorbent was simultaneously regenerated into the initial form after rinsing with water.
Awual, M. R.; 矢板 毅; 鈴木 伸一; 塩飽 秀啓
Journal of Hazardous Materials, 291, p.111 - 119, 2015/06
被引用回数:261 パーセンタイル:98.82(Engineering, Environmental)This work reports the selenium (Se(IV)) detection and removal from water by ligand functionalized organic-inorganic based novel composite adsorbent. The adsorbent exhibited distinct color change in the presence of various concentrations of Se(IV). This was characterized by UV-vis spectroscopy, and the color change was observed by naked-eye observation. The data fitted well to the Langmuir isotherm model, and the maximum Se(IV) sorption capacity was 111.12 mg/g. The presence of diverse competing ions did not affect the Se(IV) sorption capacity, and the adsorbent had almost no sorption capacity for these coexisting ions, which suggests the high selectivity to Se(IV) ions.
Awual, M. R.
Chemical Engineering Journal, 266, p.368 - 375, 2015/04
被引用回数:714 パーセンタイル:99.93(Engineering, Environmental)The soft donor atoms containing organic ligand was prepared and then successfully immobilized onto the ordered nanomaterials to form facial adsorbent for copper(II) ions removal from water. The soft donor atoms of this ligand are actively working to make a stable complex formation with Cu(II) at a neutral pH region. It is also noted that the organic ligand was acted as receptors and able to transfer chemical information into analytical signals by charge transfer transudation during the detection operation.
Awual, M. R.; Hasan, M. M.*; Shahat, A.*; Naushad, M.*; 塩飽 秀啓; 矢板 毅
Chemical Engineering Journal, 265, p.210 - 218, 2015/04
被引用回数:300 パーセンタイル:99.39(Engineering, Environmental)In this study, the organic ligand of 4-dodecyl-6-((4-(hexyloxy)phenyl)diazenyl) benzene-1,3-diol (DPDB) and mesoporous silica have been used to prepare the nano-composite adsorbent. The nano-composite adsorbent was fabricated by indirect immobilization of DPDB onto the mesoporous silica. The experimental data clarified that the Ce(III) was detected and adsorbed by the adsorbent at pH 2.50. The limit of detection for Ce(III) ions by the adsorbent was 0.12 microgram/L and the sorption capacity was 150.37 mg/g at optimum conditions. The proposed adsorbent allowed the sensitive, selective, easy to use, cost-effective, high efficiency, fast kinetics and stable capturing of Ce(III) ions even in the presence of diverse competing ions.
Awual, M. R.; Hasan, M. M.*; Znad, H.*
Chemical Engineering Journal, 259, p.611 - 619, 2015/01
被引用回数:313 パーセンタイル:99.46(Engineering, Environmental)The functional group containing organic ligand was developed and then successfully anchored onto mesoperous silica for the preparation of nano-conjugate adsorbent. After fabrication, the ligand kept open functionality for capturing Pd(II) under optimum conditions. The adsorbent exhibited the distinct color formation after adding the Pd(II) ions both in solid and liquid states. The base metal of Cu(II) and Zn(II) did not hamper the Pd(II) sorption ability of adsorbent in the acidic pH region. Therefore, it was expected that the Pd(II) could be separated from other hard metal ions by the adsorbent. Therefore the proposed adsorbent can be considered as a potential candidate for Pd(II) capturing from waste samples.