Activation mechanism of thrombopoietin receptor investigated by its specific ligand and neutralization antibodies
特異的リガンドと中和抗体を用いたトロンボポエチン受容体活性化機構の解明
松本 富美子; 安達 基泰; 清水 瑠美; 目黒 瑞枝; 新井 栄揮; 玉田 太郎; 加藤 尚志; 黒木 良太
Matsumoto, Fumiko; Adachi, Motoyasu; Shimizu, Rumi; Meguro, Mizue; Arai, Shigeki; Tamada, Taro; Kato, Takashi; Kuroki, Ryota
Thrombopoietin (TPO) is a glycoprotein that regulates the production of platelets. It stimulates the production and differentiation of megakaryocytes, the bone marrow cells that fragment into platelets. The extracellular domain of thrombopoietin receptor (ecTPOR) contains a repeat of two cytokine receptor homologous regions, CRH-1 and CRH-2. In order to investigate the activation mechanism of thrombopoietin receptor, we prepared both CRH-1 and 2 by expressing in 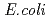 . followed by refolding, and the interaction with its specific ligand, TPO, agonistic antibody (Ab-1), and neutralization antibodies (Ab-2 and Ab-3) were investigated. Our results suggested that TPO binding to CRH-1 promote receptor dimerization. It was also suggested that CRH-2 association is important for receptor activation because the binding of Ab-2 and Ab-3 to CRH-2 resulted in neutrallization of TPO induced receptor signal transduction.
. followed by refolding, and the interaction with its specific ligand, TPO, agonistic antibody (Ab-1), and neutralization antibodies (Ab-2 and Ab-3) were investigated. Our results suggested that TPO binding to CRH-1 promote receptor dimerization. It was also suggested that CRH-2 association is important for receptor activation because the binding of Ab-2 and Ab-3 to CRH-2 resulted in neutrallization of TPO induced receptor signal transduction.