Observation of dihydrogen bonds in high-pressure phases of ammonia borane by X-ray and neutron diffraction measurements
X線と中性子回折によるアンモニアボランの高圧相中の二水素結合の観察
中野 智志*; 佐野 亜沙美 

 ; 服部 高典
; 服部 高典 

 ; 町田 真一*; 小松 一生*; 藤久 裕司*; 山脇 浩*; 後藤 義人*; 亀卦川 卓美*
; 町田 真一*; 小松 一生*; 藤久 裕司*; 山脇 浩*; 後藤 義人*; 亀卦川 卓美*
Nakano, Satoshi*; Sano, Asami; Hattori, Takanori; Machida, Shinichi*; Komatsu, Kazuki*; Fujihisa, Hiroshi*; Yamawaki, Hiroshi*; Goto, Yoshito*; Kikegawa, Takumi*
アンモニアボランのX線および中性子回折実験を常圧及び高圧下で行った。常圧下で二水素結合中のH-H距離は、ファンデルワールス半径の2倍(2.4 )よりも短い。約1.2GPaでの常圧相から第一高圧相への相転移で、半分の水素は原子間距離が延び、二水素結合が壊れた。さらに加圧すると、すべての原子間距離は2.4
)よりも短い。約1.2GPaでの常圧相から第一高圧相への相転移で、半分の水素は原子間距離が延び、二水素結合が壊れた。さらに加圧すると、すべての原子間距離は2.4 よりも短くなり、二水素結合が再び形成された。さらに、約11GPaで、空間群
よりも短くなり、二水素結合が再び形成された。さらに、約11GPaで、空間群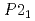 (Z=2)を持つ第二高圧相(HP2)へ転移した。この時、分子軸はより傾き、二水素結合の種類は6から11へと増えた。第3の相転移の直前(18.9GPa)で、最短の二水素結合は1.65
(Z=2)を持つ第二高圧相(HP2)へ転移した。この時、分子軸はより傾き、二水素結合の種類は6から11へと増えた。第3の相転移の直前(18.9GPa)で、最短の二水素結合は1.65 になった。本研究は、高圧下の構造相転移によりアンモニアボラン中の二水素結合が破壊、再形成するのを実験的に初めて観測したものである。
になった。本研究は、高圧下の構造相転移によりアンモニアボラン中の二水素結合が破壊、再形成するのを実験的に初めて観測したものである。
X-ray and neutron diffraction analyses of ammonia borane were conducted at ambient and high pressures. The H-H distance in dihydrogen bonds was shorter than twice the van der Waals radius (2.4  ). The half of the dihydrogen bonds were broken on phase transition from AP to the first high pressure phase (HP1) at approximately 1.2 GPa as revealed by an increase in the H-H distances. On further pressure increase, all of the H-H distances became shorter than 2.4
). The half of the dihydrogen bonds were broken on phase transition from AP to the first high pressure phase (HP1) at approximately 1.2 GPa as revealed by an increase in the H-H distances. On further pressure increase, all of the H-H distances became shorter than 2.4  again, implying the pressure-induced reformation of the dihydrogen bonds. Furthermore, the HP1 transformed to the second one with the structure of
again, implying the pressure-induced reformation of the dihydrogen bonds. Furthermore, the HP1 transformed to the second one with the structure of 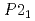 (Z = 2) at about 11 GPa. In this phase transition, the inclination of the molecule axis became larger and the number of types of dihydrogen bonds increased from 6 to 11. Just before the third transition at 18.9 GPa, the shortest dihydrogen bond decreased to 1.65
(Z = 2) at about 11 GPa. In this phase transition, the inclination of the molecule axis became larger and the number of types of dihydrogen bonds increased from 6 to 11. Just before the third transition at 18.9 GPa, the shortest dihydrogen bond decreased to 1.65  . The present study experimentally first confirmed the breakage and reformation of the dihydrogen bonds by the structural change under pressure.
. The present study experimentally first confirmed the breakage and reformation of the dihydrogen bonds by the structural change under pressure.